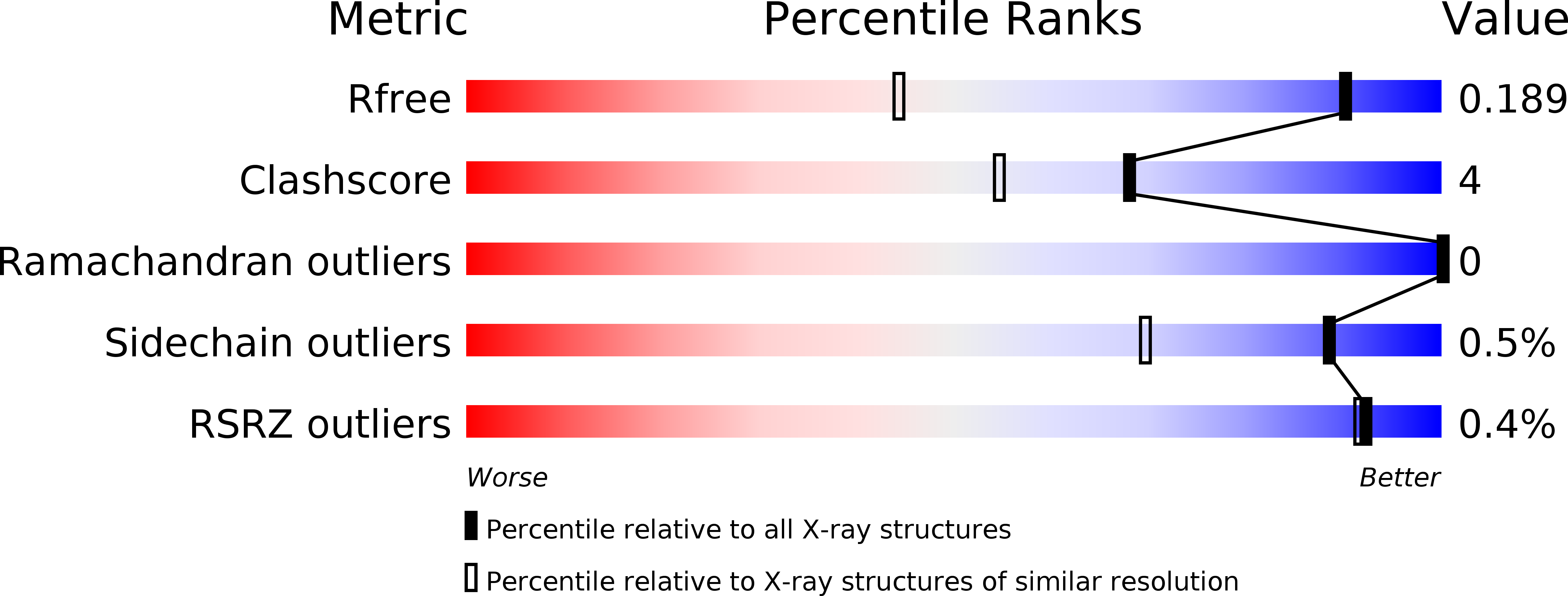
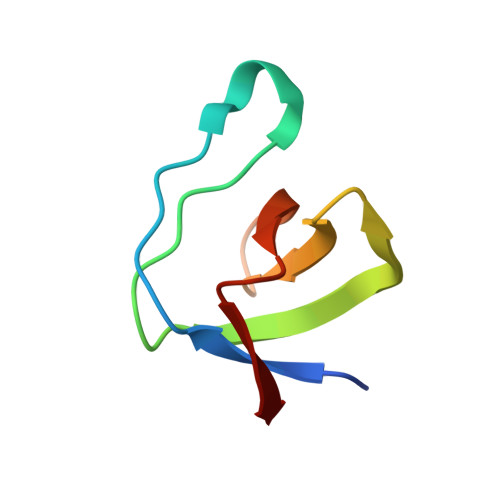
Structural insights into binding of STAC proteins to voltage-gated calcium channels.
Wong King Yuen, S.M., Campiglio, M., Tung, C.C., Flucher, B.E., Van Petegem, F.(2017) Proc Natl Acad Sci U S A 114: E9520-E9528
- PubMed: 29078335
- DOI: https://doi.org/10.1073/pnas.1708852114
- Primary Citation of Related Structures:
6B25, 6B26, 6B27, 6B28, 6B29 - PubMed Abstract:
Excitation-contraction (EC) coupling in skeletal muscle requires functional and mechanical coupling between L-type voltage-gated calcium channels (Ca V 1.1) and the ryanodine receptor (RyR1). Recently, STAC3 was identified as an essential protein for EC coupling and is part of a group of three proteins that can bind and modulate L-type voltage-gated calcium channels. Here, we report crystal structures of tandem-SH3 domains of different STAC isoforms up to 1.2-Å resolution. These form a rigid interaction through a conserved interdomain interface. We identify the linker connecting transmembrane repeats II and III in two different Ca V isoforms as a binding site for the SH3 domains and report a crystal structure of the complex with the STAC2 isoform. The interaction site includes the location for a disease variant in STAC3 that has been linked to Native American myopathy (NAM). Introducing the mutation does not cause misfolding of the SH3 domains, but abolishes the interaction. Disruption of the interaction via mutations in the II-III loop perturbs skeletal muscle EC coupling, but preserves the ability of STAC3 to slow down inactivation of Ca V 1.2.
Organizational Affiliation:
Department of Biochemistry and Molecular Biology, University of British Columbia, V6T 1Z3 Vancouver, BC, Canada.