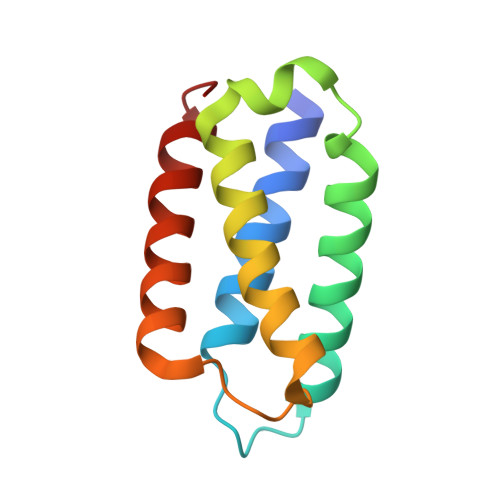
Crystal structure of Psb27 from Arabidopsis thaliana determined at a resolution of 1.85 angstrom.
Cheng, X., Liu, J., Huan, Z., Fudong, L., Shuya, Z., Min, X., Ke, R., Yuhua, W., Aigen, F.(2018) Photosynth Res 136: 139-146
- PubMed: 29098572
- DOI: https://doi.org/10.1007/s11120-017-0450-3
- Primary Citation of Related Structures:
5X56 - PubMed Abstract:
Proper biogenesis and maintenance of photosynthetic thylakoid membrane complexes are essential for the photosynthetic light reactions. A thylakoid lumenal protein, Psb27, plays a vital role in assembly or/and maintenance of photosystem II (PSII). In cyanobacteria, it is a small lipoprotein docked to the lumenal side of PSII, and functions in the assembly of the Mn 4 Ca cluster and in the PSII repair cycle. However, Psb27 from Arabidopsis thaliana is not a lipoprotein, and it is involved in PSII repair and acclimation to fluctuating light stress, suggesting a functional divergence between Arabidopsis Psb27 and cyanobacterial Psb27s. To gain a better understanding of Psb27 from higher plants, we determined the crystal structure of Arabidopsis Psb27 by X-ray crystallography at a resolution of 1.85 Å. The structure of Arabidopsis Psb27 is a four-helix bundle, similar to its orthologues from cyanobacteria. However, there are several structural differences between Arabidopsis Psb27 and cyanobacterial Psb27s concerning the overall molecular shape, the N- and C-terminal structures, and the surface charge. These differences suggest that Psb27 from higher plants and cyanobacteria may function differently.
Organizational Affiliation:
Chinese Education Ministry's Key Laboratory of Western Resources and Modern Biotechnology, Key Laboratory of Biotechnology Shaanxi Province, College of Life Sciences, Northwest University, 229 North Taibai Road, Xi'an, 710069, Shaanxi, China.