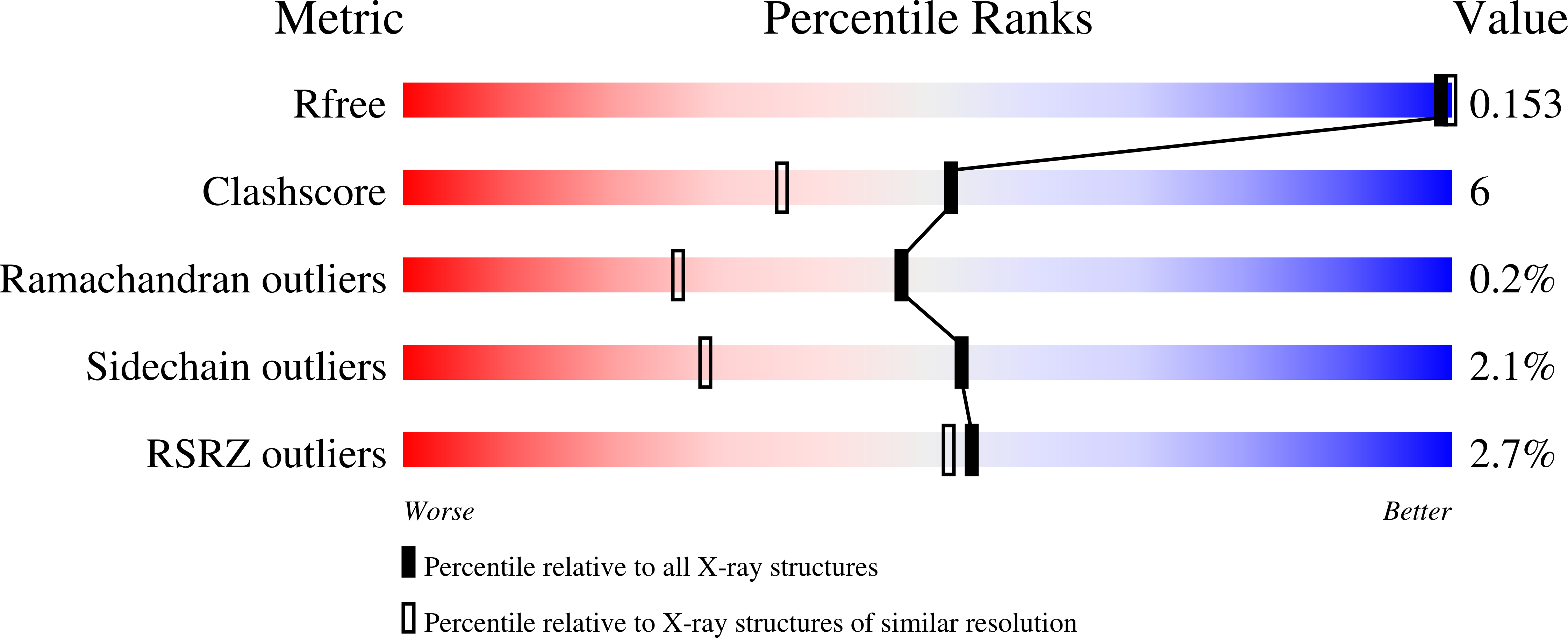
Structural Insights into the Catalytic Mechanism of Trypanosoma cruzi trans-Sialidase
Amaya, M.F., Watts, A.G., Damager, I., Wehenkel, A., Nguyen, T., Buschiazzo, A., Paris, G., Frasch, A.C., Withers, S.G., Alzari, P.M.(2004) Structure 12: 775-784
- PubMed: 15130470
- DOI: https://doi.org/10.1016/j.str.2004.02.036
- Primary Citation of Related Structures:
1S0I, 1S0J, 2AH2 - PubMed Abstract:
Sialidases are a superfamily of sialic-acid-releasing enzymes that are of significant interest due to their implication as virulence factors in the pathogenesis of a number of diseases. However, extensive studies of viral and microbial sialidases have failed to provide a comprehensive picture of their mechanistic properties, in part because the structures of competent enzyme-substrate complexes and reaction intermediates have never been described. Here we report these structures for the Trypanosoma cruzi trans-sialidase (TcTS), showing that catalysis by sialidases occurs via a similar mechanism to that of other retaining glycosidases, but with some intriguing differences that may have evolved in response to the substrate structure.
Organizational Affiliation:
Unité de Biochimie Structurale, CNRS URA 2185, Institut Pasteur, 25 rue du Dr. Roux, 75724 Paris, France.