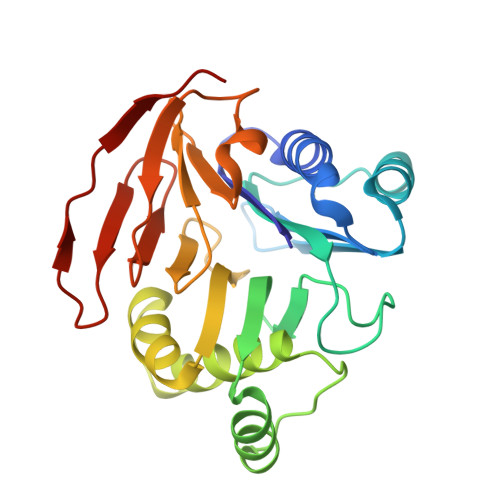
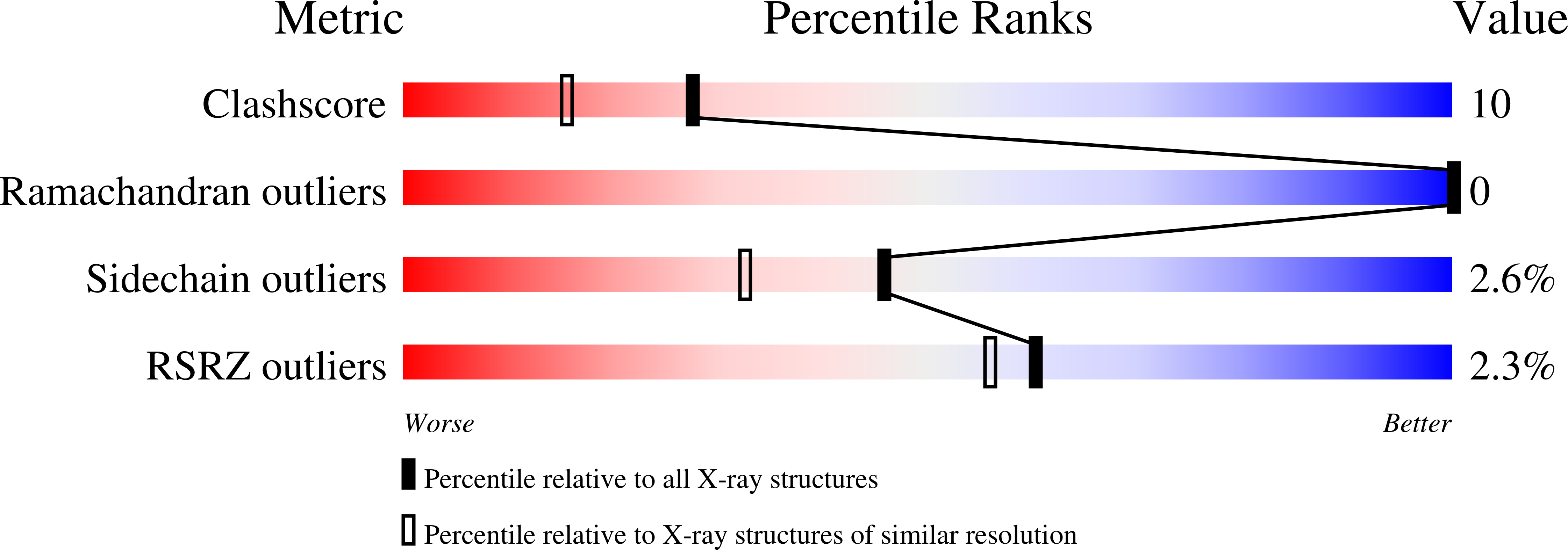Substrate Binding is Required for Assembly of the Active Conformation of the Catalytic Site in Ntn Amidotransferases: Evidence from the 1.8 Angstrom Crystal Structure of the Glutaminase Domain of Glucosamine 6-Phosphate Synthase
Isupov, M.N., Obmolova, G., Butterworth, S., Badet-Denisot, M.-A., Badet, B., Polikarpov, I., Littlechild, J.A., Teplyakov, A.(1996) Structure 4: 801-810
- PubMed: 8805567
- DOI: https://doi.org/10.1016/s0969-2126(96)00087-1
- Primary Citation of Related Structures:
1XFF, 1XFG - PubMed Abstract:
Amidotransferases use the amide nitrogen of glutamine in a number of important biosynthetic reactions. They are composed of a glutaminase domain, which catalyzes the hydrolysis of glutamine to glutamate and ammonia, and a synthetase domain, catalyzing amination of the substrate. To gain insight into the mechanism of nitrogen transfer, we examined the structure of the glutaminase domain of glucosamine 6-phosphate synthase (GLMS).
Organizational Affiliation:
Department of Chemistry and Biological Sciences, University of Exeter, UK.