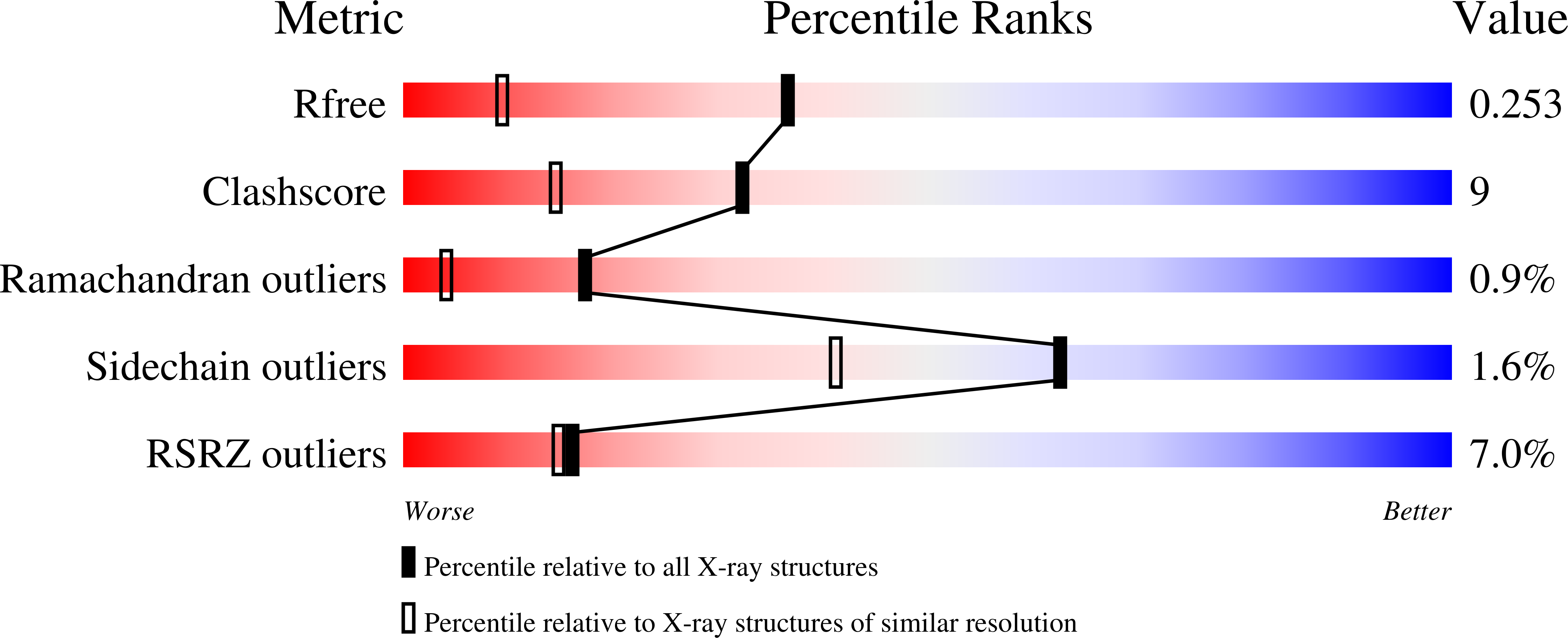
Sialic Acid Recognition by Vibrio Cholerae Neuraminidase.
Moustafa, I., Connaris, H., Taylor, M., Zaitsev, V., Wilson, J.C., Kiefel, M.J., Von Itzstein, M., Taylor, G.(2004) J Biol Chem 279: 40819
- PubMed: 15226294
- DOI: https://doi.org/10.1074/jbc.M404965200
- Primary Citation of Related Structures:
1W0O, 1W0P - PubMed Abstract:
Vibrio cholerae neuraminidase (VCNA) plays a significant role in the pathogenesis of cholera by removing sialic acid from higher order gangliosides to unmask GM1, the receptor for cholera toxin. We previously showed that the structure of VCNA is composed of a central beta-propeller catalytic domain flanked by two lectin-like domains; however the nature of the carbohydrates recognized by these lectin domains has remained unknown. We present here structures of the enzyme in complex with two substrates, alpha-2,3-sialyllactose and alpha-2,6-sialyllactose. Both substrate complexes reveal the alpha-anomer of N-acetylneuraminic acid (Neu5Ac) bound to the N-terminal lectin domain, thereby revealing the role of this domain. The large number of interactions suggest a relatively high binding affinity for sialic acid, which was confirmed by calorimetry, which gave a Kd approximately 30 microm. Saturation transfer difference NMR using a non-hydrolyzable substrate, Neu5,9Ac2-2-S-(alpha-2,6)-GlcNAcbeta1Me, was also used to map the ligand interactions at the VCNA lectin binding site. It is well known that VCNA can hydrolyze both alpha-2,3- and alpha-2,6-linked sialic acid substrates. In this study using alpha-2,3-sialyllactose co-crystallized with VCNA it was revealed that the inhibitor 2-deoxy-2,3-didehydro-N-acetylneuraminic acid (Neu5Ac2en) was bound at the catalytic site. This observation supports the notion that VCNA can produce its own inhibitor and has been further confirmed by 1H NMR analysis. The discovery of the sialic acid binding site in the N-lectin-like domain suggests that this might help target VCNA to sialic acid-rich environments, thereby enhancing the catalytic efficiency of the enzyme.
Organizational Affiliation:
Centre for Biomolecular Sciences, University of St. Andrews, St. Andrews, Fife KY16 9ST, Scotland.