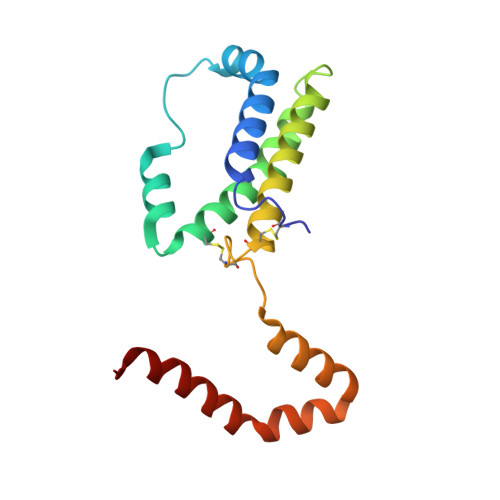
Crystal structure of interleukin-10 reveals the functional dimer with an unexpected topological similarity to interferon gamma.
Zdanov, A., Schalk-Hihi, C., Gustchina, A., Tsang, M., Weatherbee, J., Wlodawer, A.(1995) Structure 3: 591-601
- PubMed: 8590020
- DOI: https://doi.org/10.1016/s0969-2126(01)00193-9
- Primary Citation of Related Structures:
1ILK - PubMed Abstract:
Interleukin (IL)-10 is a cytokine that inhibits production of other regulatory factors, including interferon gamma (IFN-gamma) and IL-2. A dimer of IL-10 is present in solution and is presumed to participate in receptor binding, but the nature of the dimer has not been previously reported. An atomic model is necessary to interpret biological activity of IL-10 and to design mutants with agonistic or antagonistic properties. The X-ray crystal structure of a recombinant form of human IL-10 has been solved at 1.8 A resolution and refined to a crystallographic R-factor of 0.156. The molecule is a tight dimer made of two interpenetrating subunits, forming a V-shaped structure. Each half of the structure consists of a six alpha-helices, four originating from one subunit and two from the other. Four of the helices form a classical 'up-up-down-down' bundle observed in all other helical cytokines. The overall topology of the helices bears close resemblance to IFN gamma, although the similarity is less striking when examined in greater detail. The topological similarity of IL-10 to IFN gamma was totally unexpected, and may be a reflection of the close relationship between the biological effects of these two cytokines. The structure of IL-10 provides insights into the possible modes of conversion of the dimer into monomers, and of putative sites of receptor interactions. The good level of refinement and high resolution of this structure show that the internal disorder often associated with other helical cytokines is not an essential feature of this class of proteins.
Organizational Affiliation:
Macromolecular Structure Laboratory, NCI-Frederick Cancer Research and Development Center, Maryland 21702, USA.