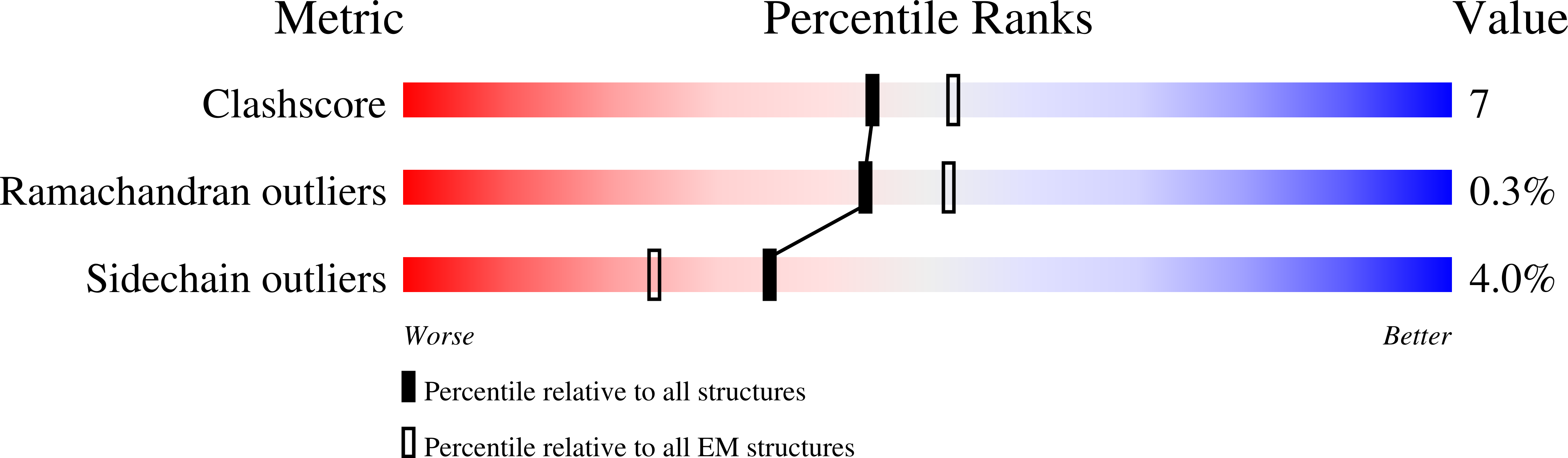Cryo-EM structures of anti-malarial antibody L9 with circumsporozoite protein reveal trimeric L9 association and complete 27-residue epitope.
Tripathi, P., Bender, M.F., Lei, H., Da Silva Pereira, L., Shen, C.H., Bonilla, B., Dillon, M., Ou, L., Pancera, M., Wang, L.T., Zhang, B., Batista, F.D., Idris, A.H., Seder, R.A., Kwong, P.D.(2023) Structure 31: 480-491.e4
- PubMed: 36931276
- DOI: https://doi.org/10.1016/j.str.2023.02.009
- Primary Citation of Related Structures:
8EK1, 8EKA - PubMed Abstract:
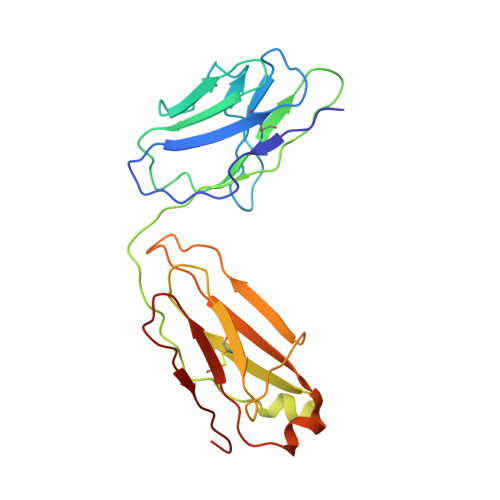
Monoclonal antibody L9 recognizes the Plasmodium falciparum circumsporozoite protein (PfCSP) and is highly protective following controlled human malaria challenge. To gain insight into its function, we determined cryoelectron microscopy (cryo-EM) structures of L9 in complex with full-length PfCSP and assessed how this recognition influenced protection by wild-type and mutant L9s. Cryo-EM reconstructions at 3.6- and 3.7-Å resolution revealed L9 to recognize PfCSP as an atypical trimer. Each of the three L9s in the trimer directly recognized an Asn-Pro-Asn-Val (NPNV) tetrapeptide on PfCSP and interacted homotypically to facilitate L9-trimer assembly. We analyzed peptides containing different repeat tetrapeptides for binding to wild-type and mutant L9s to delineate epitope and homotypic components of L9 recognition; we found both components necessary for potent malaria protection. Last, we found the 27-residue stretch recognized by L9 to be highly conserved in P. falciparum isolates, suggesting the newly revealed complete L9 epitope to be an attractive vaccine target.
Organizational Affiliation:
Vaccine Research Center, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, MD 20892, USA.