Crystal Structure of FolP (dihydropteroate synthase) from Colstridium difficile in the presence of pteroic acid
Girardi, N.M., Thoden, J.B., Holden, H.M.To be published.
Experimental Data Snapshot
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Dihydropteroate synthase | 267 | Clostridioides difficile | Mutation(s): 0 Gene Names: folP, SAMEA3374989_02220 EC: 2.5.1.15 | 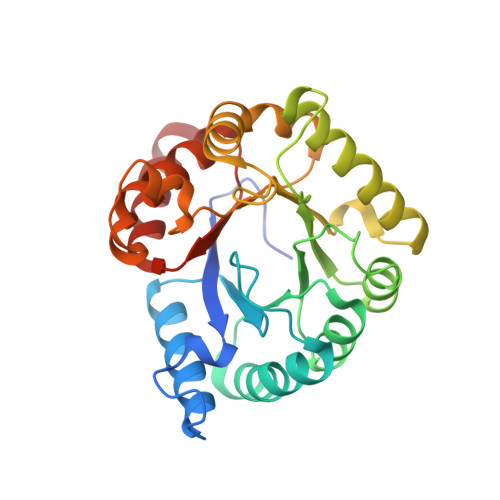 | |
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| PT1 (Subject of Investigation/LOI) Query on PT1 | K [auth A] | PTEROIC ACID C14 H12 N6 O3 JOAQINSXLLMRCV-UHFFFAOYSA-N |  | ||
| NHE Query on NHE | L [auth A] | 2-[N-CYCLOHEXYLAMINO]ETHANE SULFONIC ACID C8 H17 N O3 S MKWKNSIESPFAQN-UHFFFAOYSA-N |  | ||
| SO4 Query on SO4 | B [auth A] C [auth A] D [auth A] E [auth A] F [auth A] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 156.258 | α = 90 |
| b = 156.258 | β = 90 |
| c = 84.708 | γ = 120 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| PDB_EXTRACT | data extraction |
| HKL-3000 | data reduction |
| HKL-3000 | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | gm115921 |