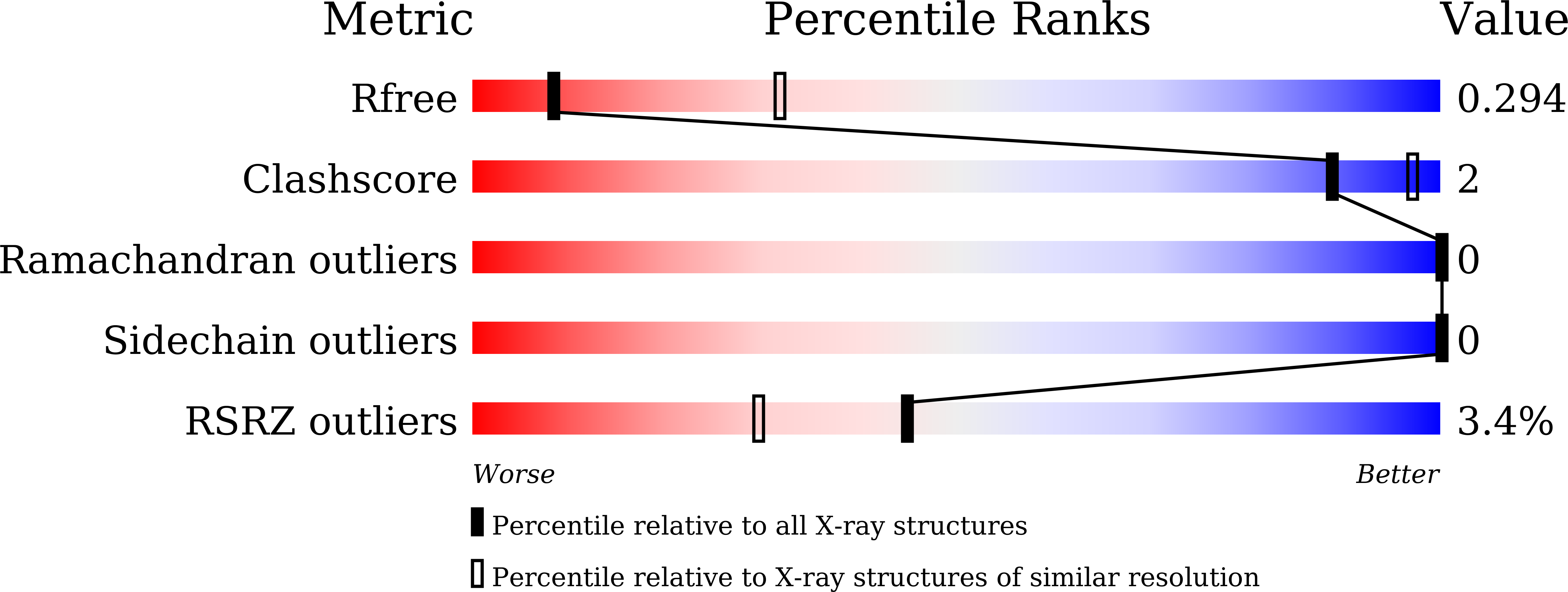
The crystal structure of sulfiredoxin from Arabidopsis thaliana revealed a more robust antioxidant mechanism in plants.
Liu, M., Wang, J., Li, X., Sylvanno, M.J., Li, M., Zhang, M., Wang, M.(2019) Biochem Biophys Res Commun 520: 347-352
- PubMed: 31604522
- DOI: https://doi.org/10.1016/j.bbrc.2019.10.034
- Primary Citation of Related Structures:
6KY4 - PubMed Abstract:
Typical 2-cysteine peroxiredoxins (2-Cys Prxs) are critical peroxidase sensors and could be deactivated by the hyperoxidation under oxidative stress. In plants, 2-Cys Prxs present at a high level in chloroplasts and are repaired by Sulfiredoxin. Whereas many studies have explored the mechanism of Sulfiredoxin from Homo sapiens (HsSrx), the molecular mechanism of Sulfiredoxin in plants with unique photosynthesis remains unclear. Here we report the crystal structure of Sulfiredoxin from Arabidopsis thaliana (AtSrx), which displayed a typical ParB/Srx fold with an ATP bound at a conservative nucleotide binding motif GCHR. Both the ADP binding pocket and the putative AtSrx-AtPrxA interaction surface of AtSrx are more positively charged comparing to HsSrx, suggesting a robust mechanism of AtSrx. These features illustrate the unique mechanisms of AtSrx, which are vital for figure out the strategies of plants to cope with oxidation stress.
Organizational Affiliation:
School of Life Sciences, Anhui University, Hefei, Anhui, 230601, China; Institutes of Physical Science and Information Technology, Anhui University, Hefei, Anhui, 230601, China; Key Laboratory of Human Microenvironment and Precision Medicine of Anhui Higher Education Institutes, Anhui University, Hefei, Anhui, 230601, China.