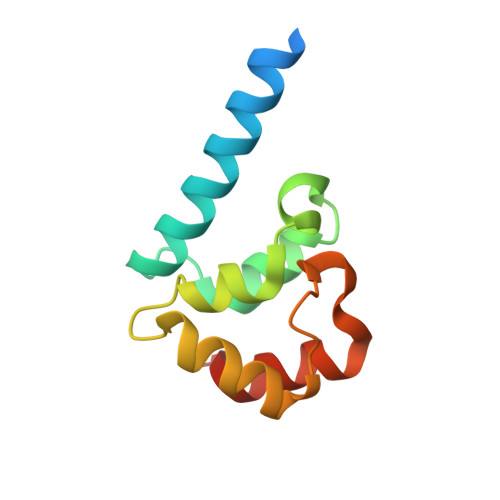
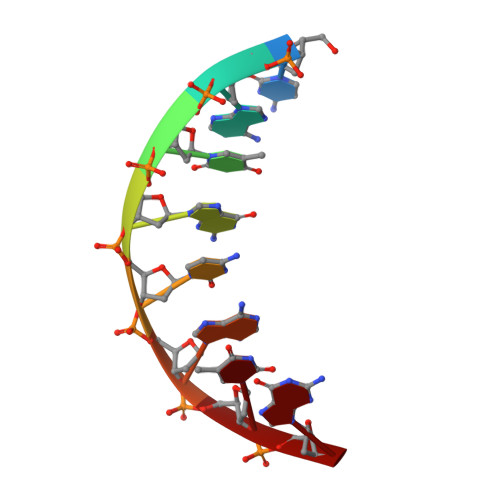
Streptomyces IHF uses multiple interfaces to bind DNA.
Nanji, T., Gehrke, E.J., Shen, Y., Gloyd, M., Zhang, X., Firby, C.D., Huynh, A., Razi, A., Ortega, J., Elliot, M.A., Guarne, A.(2019) Biochim Biophys Acta Gen Subj 1863: 129405-129405
- PubMed: 31376411
- DOI: https://doi.org/10.1016/j.bbagen.2019.07.014
- Primary Citation of Related Structures:
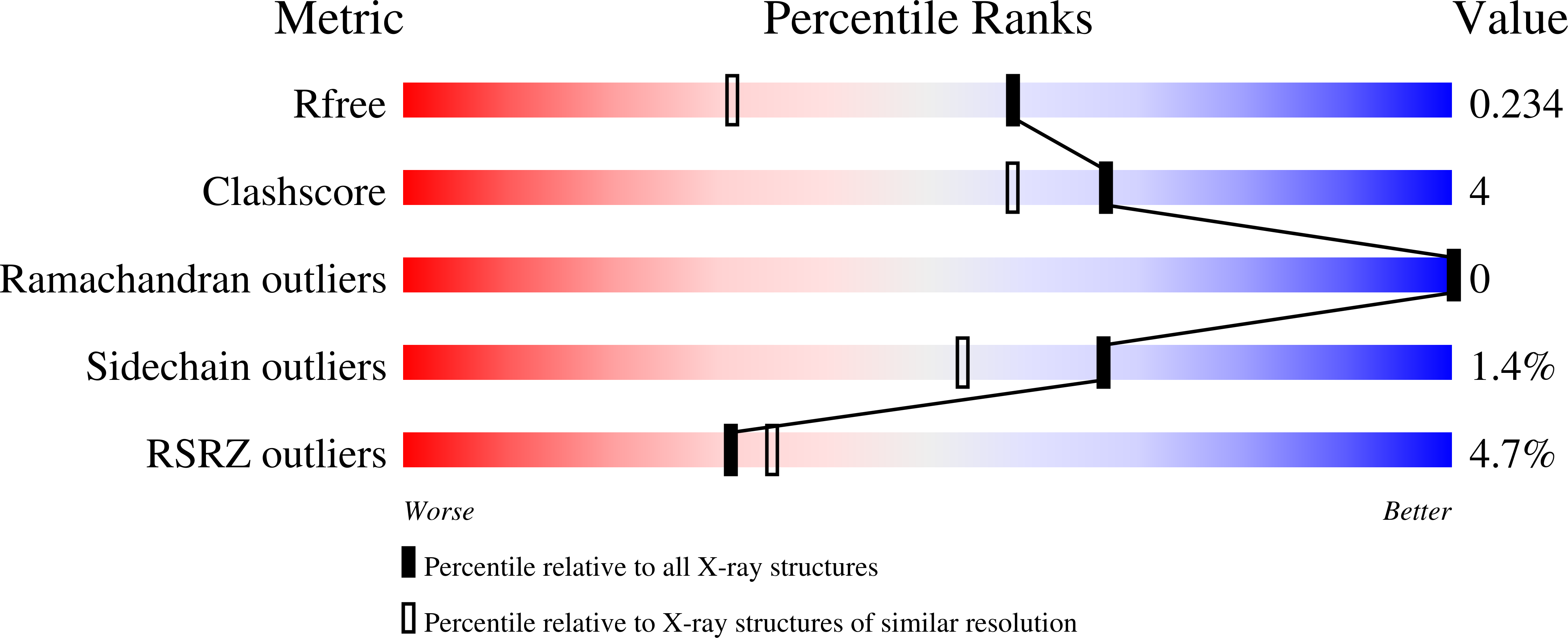
6BEK - PubMed Abstract:
Nucleoid associated proteins (NAPs) are essential for chromosome condensation in bacterial cells. Despite being a diverse group, NAPs share two common traits: they are small, oligomeric proteins and their oligomeric state is critical for DNA condensation. Streptomyces coelicolor IHF (sIHF) is an actinobacterial-specific nucleoid-associated protein that despite its name, shares neither sequence nor structural homology with the well-characterized Escherichia coli IHF. Like E. coli IHF, sIHF is needed for efficient nucleoid condensation, morphological development and antibiotic production in S. coelicolor.
Organizational Affiliation:
Department of Biochemistry and Biomedical Sciences, McMaster University, Hamilton, ON, Canada.