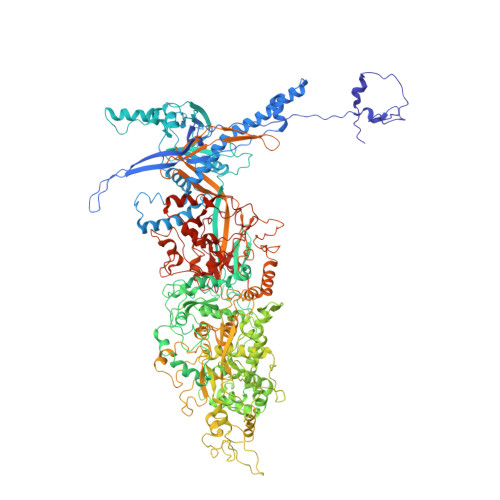
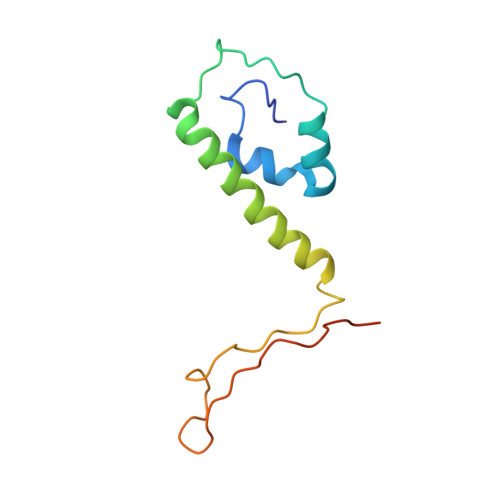
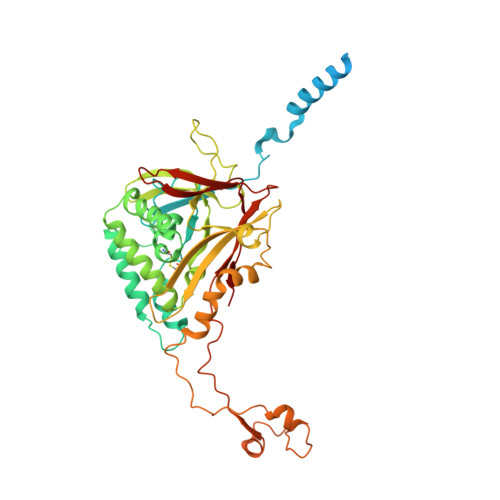
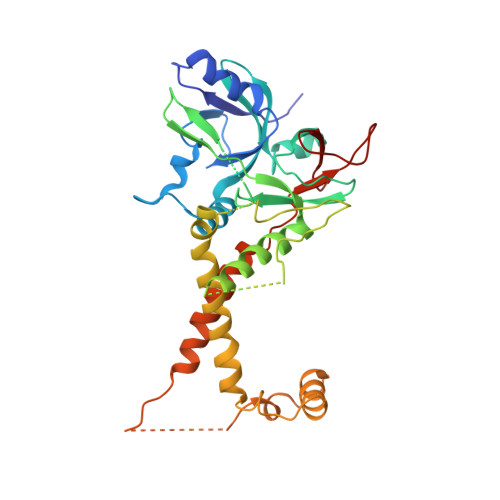
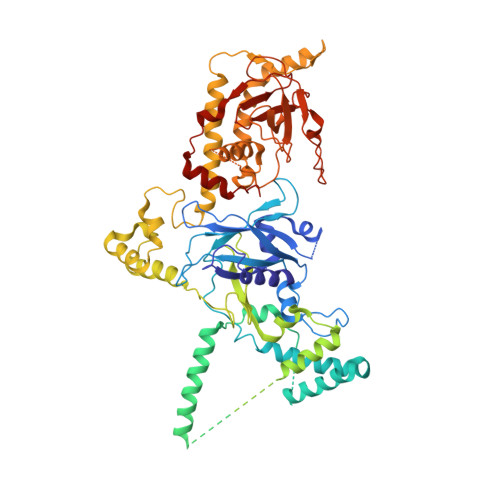
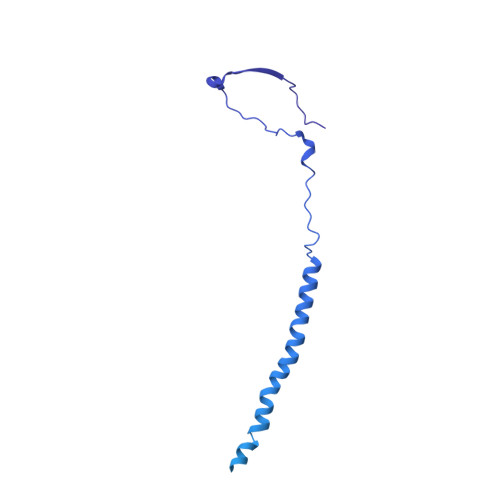
Cryo-EM structures of herpes simplex virus type 1 portal vertex and packaged genome.
Liu, Y.T., Jih, J., Dai, X., Bi, G.Q., Zhou, Z.H.(2019) Nature 570: 257-261
- PubMed: 31142842
- DOI: https://doi.org/10.1038/s41586-019-1248-6
- Primary Citation of Related Structures:
6OD7, 6ODM - PubMed Abstract:
Herpesviruses are enveloped viruses that are prevalent in the human population and are responsible for diverse pathologies, including cold sores, birth defects and cancers. They are characterized by a highly pressurized pseudo-icosahedral capsid-with triangulation number (T) equal to 16-encapsidating a tightly packed double-stranded DNA (dsDNA) genome 1-3 . A key process in the herpesvirus life cycle involves the recruitment of an ATP-driven terminase to a unique portal vertex to recognize, package and cleave concatemeric dsDNA, ultimately giving rise to a pressurized, genome-containing virion 4,5 . Although this process has been studied in dsDNA phages 6-9 -with which herpesviruses bear some similarities-a lack of high-resolution in situ structures of genome-packaging machinery has prevented the elucidation of how these multi-step reactions, which require close coordination among multiple actors, occur in an integrated environment. To better define the structural basis of genome packaging and organization in herpes simplex virus type 1 (HSV-1), we developed sequential localized classification and symmetry relaxation methods to process cryo-electron microscopy (cryo-EM) images of HSV-1 virions, which enabled us to decouple and reconstruct hetero-symmetric and asymmetric elements within the pseudo-icosahedral capsid. Here we present in situ structures of the unique portal vertex, genomic termini and ordered dsDNA coils in the capsid spooled around a disordered dsDNA core. We identify tentacle-like helices and a globular complex capping the portal vertex that is not observed in phages, indicative of herpesvirus-specific adaptations in the DNA-packaging process. Finally, our atomic models of portal vertex elements reveal how the fivefold-related capsid accommodates symmetry mismatch imparted by the dodecameric portal-a longstanding mystery in icosahedral viruses-and inform possible DNA-sequence recognition and headful-sensing pathways involved in genome packaging. This work showcases how to resolve symmetry-mismatched elements in a large eukaryotic virus and provides insights into the mechanisms of herpesvirus genome packaging.
Organizational Affiliation:
Center for Integrative Imaging, Hefei National Laboratory for Physical Sciences at the Microscale, University of Science and Technology of China, Hefei, China.