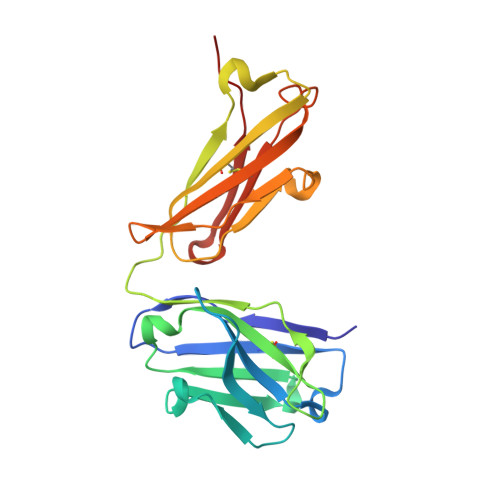
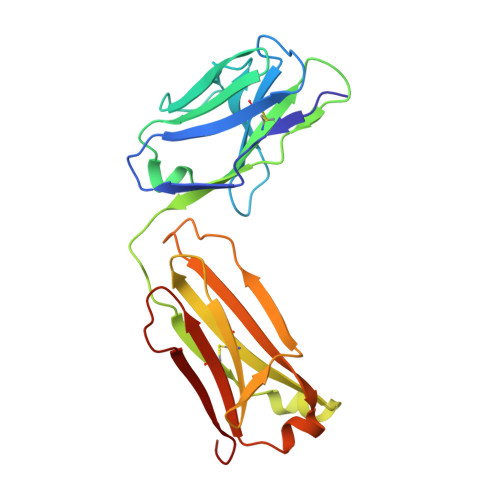
Crystal structure, epitope, and functional impact of an antibody against a superactive FVIIa provide insights into allosteric mechanism.
Jiang, L.G., Xie, X., Li, J., Persson, E., Huang, M.D.(2019) Res Pract Thromb Haemost 3: 412-419
- PubMed: 31294329
- DOI: https://doi.org/10.1002/rth2.12211
- Primary Citation of Related Structures:
5YUP - PubMed Abstract:
Blood coagulation factor VIIa (FVIIa) plays its critical physiological role in the initiation of hemostasis. Even so, recombinant FVIIa is successfully used as a bypassing agent for factor VIII or IX in the treatment of bleeds in patients with severe hemophilia with inhibitors. To investigate the utility of more potent FVIIa variants with enhanced intrinsic activity, molecules such as V21D/E154V/M156Q-FVIIa (FVIIa DVQ ) were designed.
Organizational Affiliation:
College of Chemistry National & Local Joint Biomedical Engineering Research Center on Photodynamic Technologies Fuzhou University Fuzhou China.