Ancestral mutations as a tool for solubilizing proteins: The case of a hydrophobic phosphate-binding protein.
Gonzalez, D., Hiblot, J., Darbinian, N., Miller, J.C., Gotthard, G., Amini, S., Chabriere, E., Elias, M.(2014) FEBS Open Bio 4: 121-127
- PubMed: 24490136
- DOI: https://doi.org/10.1016/j.fob.2013.12.006
- Primary Citation of Related Structures:
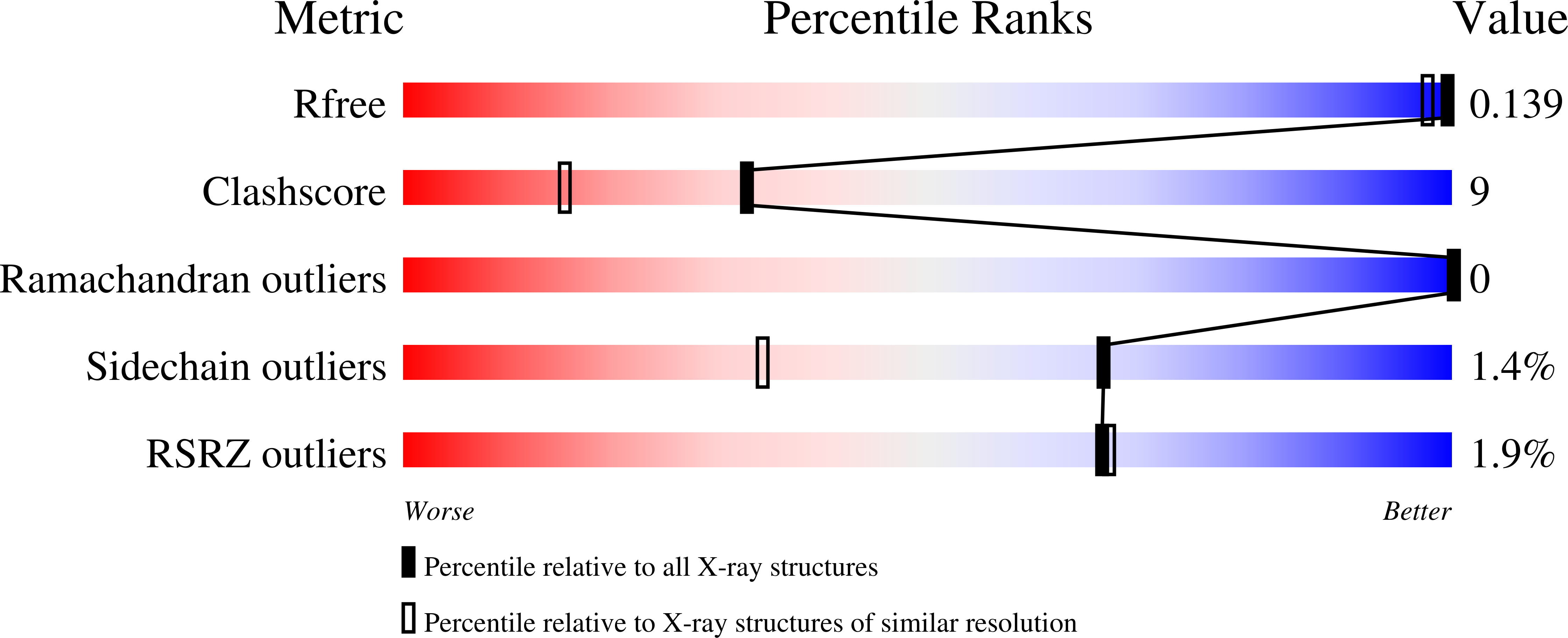
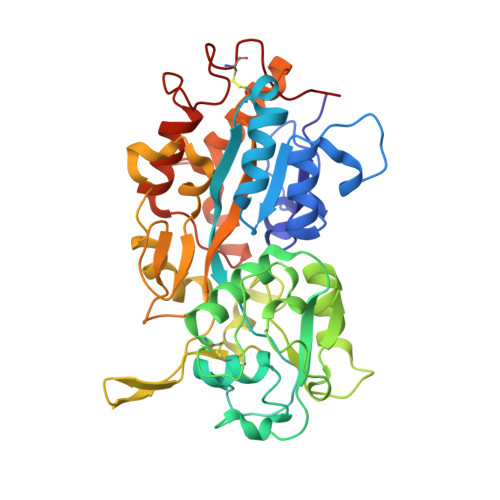
4M1V - PubMed Abstract:
Stable and soluble proteins are ideal candidates for functional and structural studies. Unfortunately, some proteins or enzymes can be difficult to isolate, being sometimes poorly expressed in heterologous systems, insoluble and/or unstable. Numerous methods have been developed to address these issues, from the screening of various expression systems to the modification of the target protein itself. Here we use a hydrophobic, aggregation-prone, phosphate-binding protein (HPBP) as a case study. We describe a simple and fast method that selectively uses ancestral mutations to generate a soluble, stable and functional variant of the target protein, here named sHPBP. This variant is highly expressed in Escherichia coli, is easily purified and its structure was solved at much higher resolution than its wild-type progenitor (1.3 versus 1.9 Å, respectively).
Organizational Affiliation:
URMITE UMR CNRS-IRD 6236, IFR48, Faculté de Médecine et de Pharmacie, Université de la Méditerranée, Marseille, France.