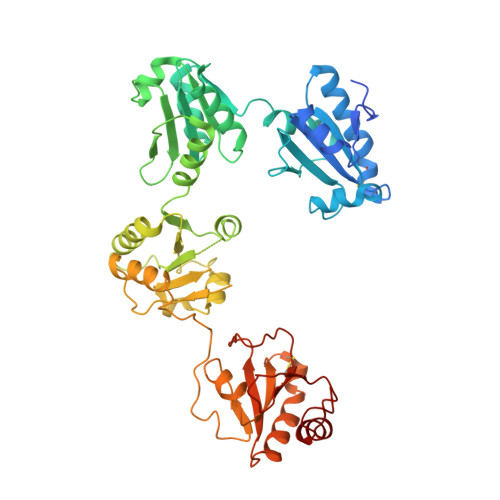
Structural insights into the redox-regulated dynamic conformations of human protein disulfide isomerase
Wang, C., Li, W., Ren, J., Fang, J., Ke, H., Gong, W., Feng, W., Wang, C.-C.(2013) Antioxid Redox Signal 19: 44-53
- PubMed: 22657537
- DOI: https://doi.org/10.1089/ars.2012.4630
- Primary Citation of Related Structures:
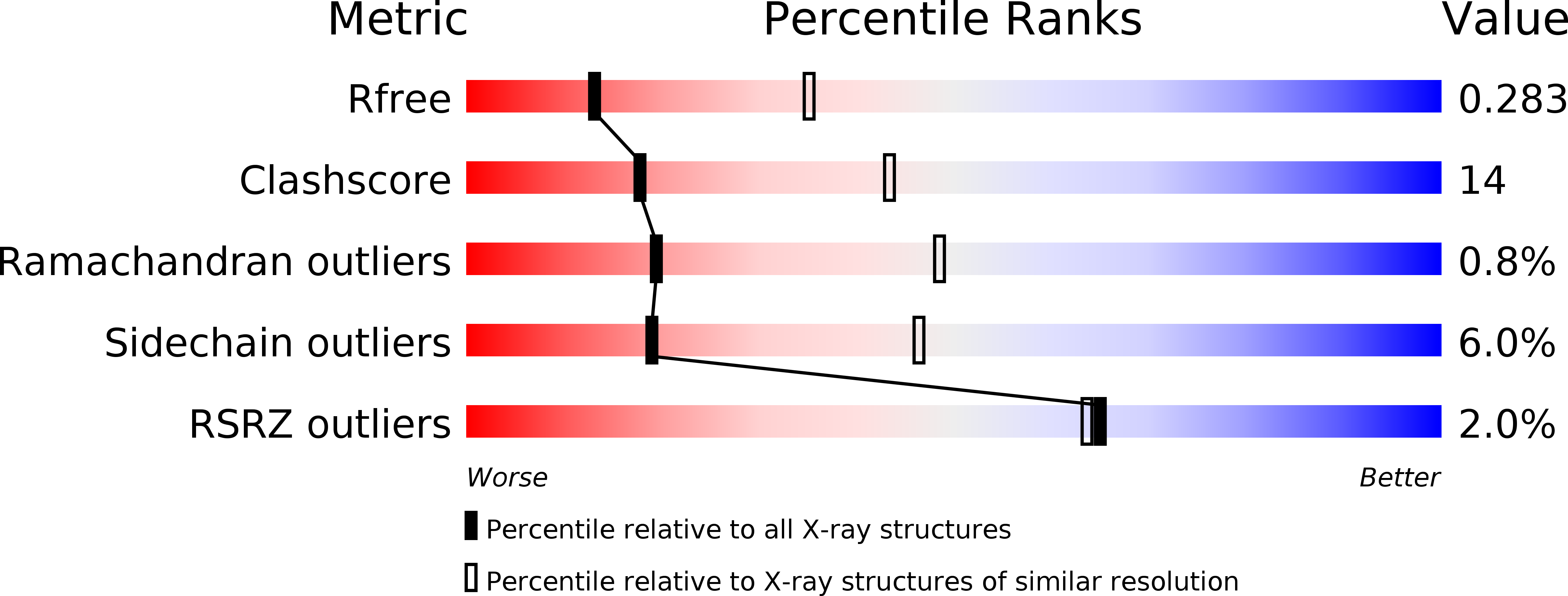
4EKZ, 4EL1 - PubMed Abstract:
Human protein disulfide isomerase (hPDI) is a key enzyme and a redox-regulated chaperone responsible for oxidative protein folding in the endoplasmic reticulum. This work aims to reveal the molecular mechanism underlying the redox-regulated functions of hPDI by determining the crystal structures of hPDI in different redox states.
Organizational Affiliation:
National Laboratory of Biomacromolecules, Institute of Biophysics, Chinese Academy of Sciences, Beijing, China.