Molecular Basis for the Association of Microcephalin (MCPH1) Protein with the Cell Division Cycle Protein 27 (Cdc27) Subunit of the Anaphase-promoting Complex.
Singh, N., Wiltshire, T.D., Thompson, J.R., Mer, G., Couch, F.J.(2012) J Biol Chem 287: 2854-2862
- PubMed: 22139841
- DOI: https://doi.org/10.1074/jbc.M111.307868
- Primary Citation of Related Structures:
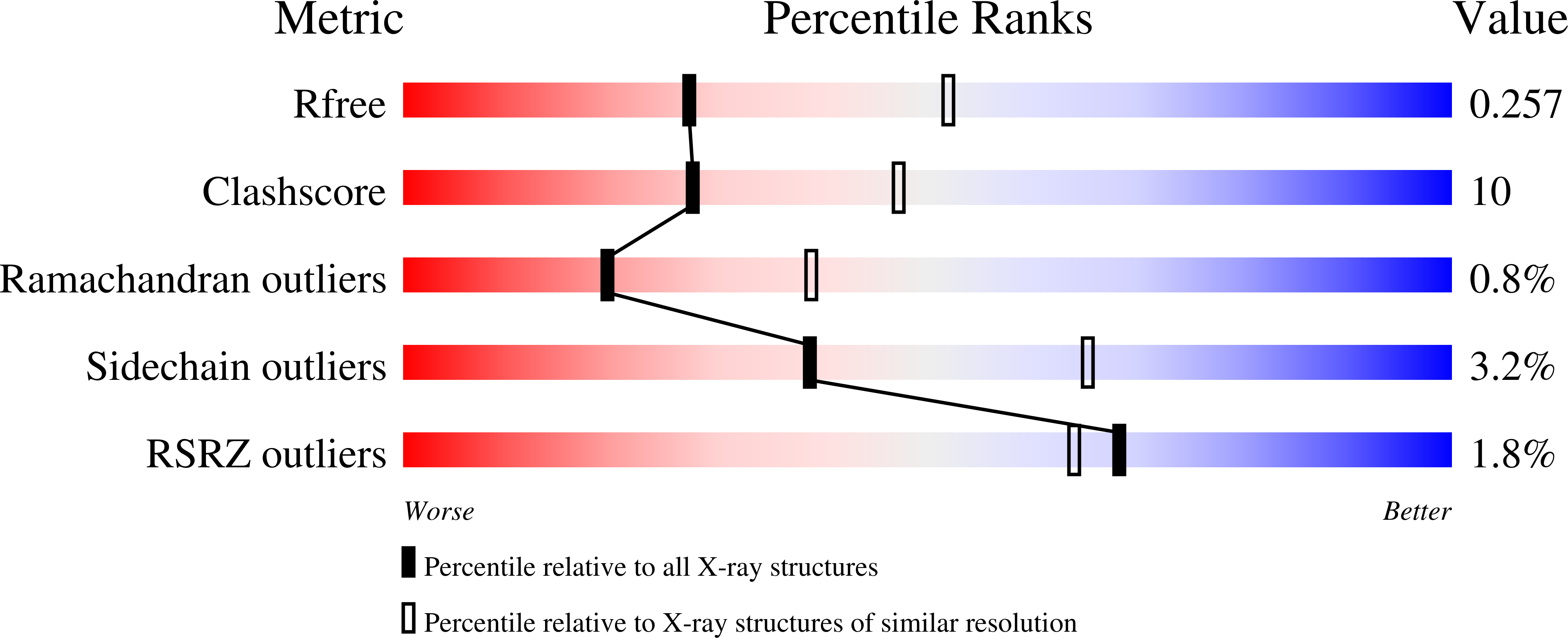
2ETX, 3T1N - PubMed Abstract:
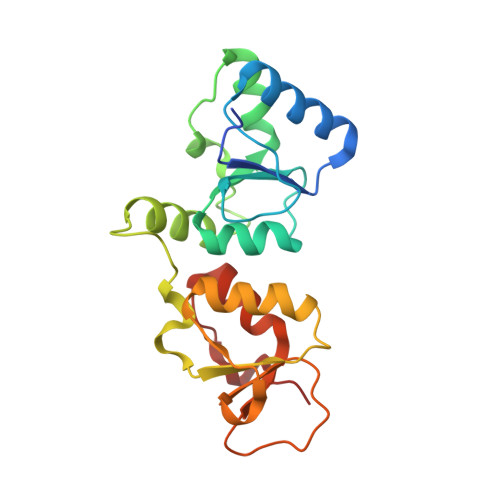
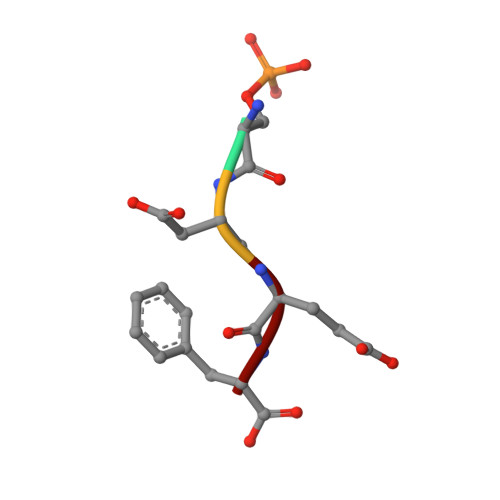
Microcephalin (MCPH1), the first gene identified as causative for primary recessive autosomal microcephaly, is aberrantly expressed in autism-like disorders and human malignancy of breast and ovarian origin. MCPH1, the encoded protein product, has been implicated in various cellular processes including the DNA damage checkpoint, DNA repair, and transcription. Although our understanding of the cellular context in which MCPH1 operates continues to develop, a structural understanding of the C-terminal tandem BRCT domains of MCPH1 remains unexplored. Here, we identify cell division cycle protein 27 (Cdc27), a component of the anaphase-promoting complex (APC/C), as a novel interacting partner of MCPH1. We provide in vitro and in vivo evidence that the C-terminal tandem BRCT domains of MCPH1 (C-BRCTs) bind Cdc27 in a phosphorylation-dependent manner. To characterize this interaction further, we determined the structure of MCPH1 C-BRCTs in complex with a phosphorylated Cdc27 peptide (pCdc27) using x-ray crystallography. Based on this structure, we identified single amino acid mutations targeted at the binding interface that disrupted the MCPH1-pCdc27 interaction. Collectively, our data define the biochemical, structural, and cellular determinants of the novel interaction between MCPH1 and Cdc27 and suggest that this interaction may occur within the larger context of MCPH1-APC/C.
Organizational Affiliation:
Department of Biochemistry and Molecular Biology, Mayo Clinic, Rochester, Minnesota 55905, USA.