Crystal Structure of a Coiled-Coil Domain from Human ROCK I.
Tu, D., Li, Y., Song, H.K., Toms, A.V., Gould, C.J., Ficarro, S.B., Marto, J.A., Goode, B.L., Eck, M.J.(2011) PLoS One 6: e18080-e18080
- PubMed: 21445309
- DOI: https://doi.org/10.1371/journal.pone.0018080
- Primary Citation of Related Structures:
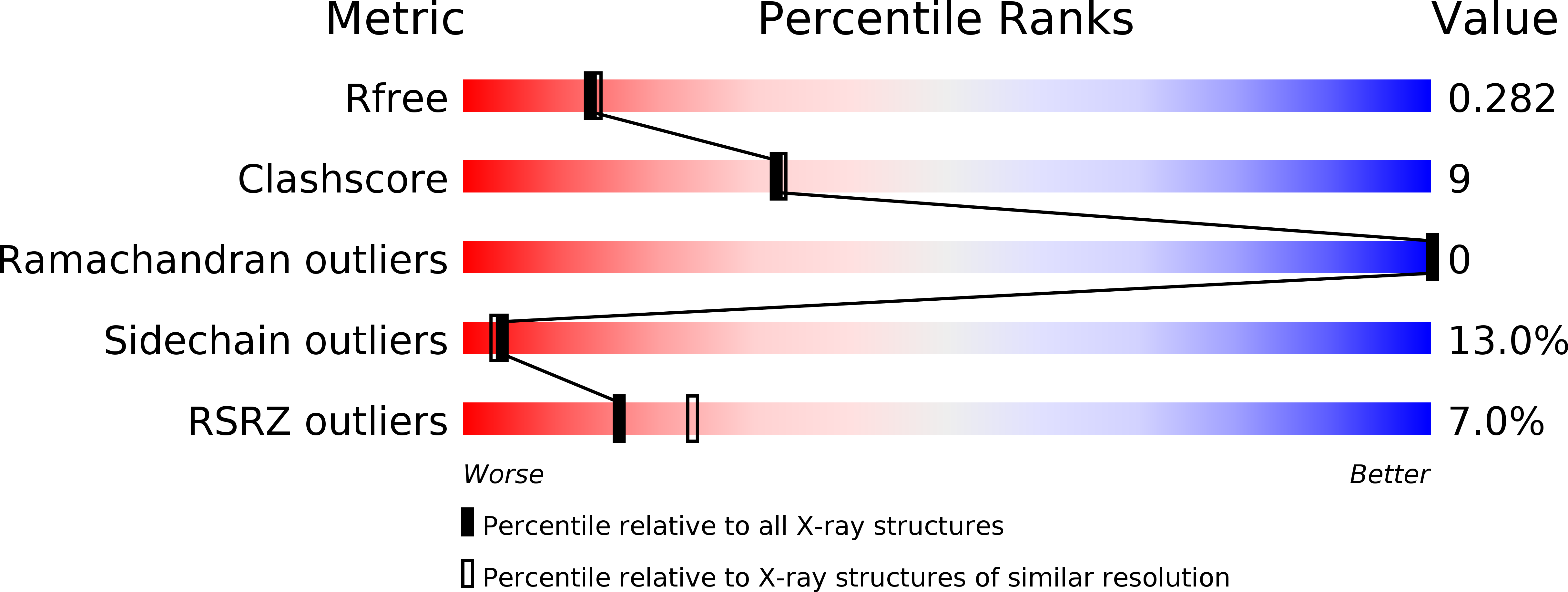
3O0Z - PubMed Abstract:
The small GTPase Rho and one of its targets, Rho-associated kinase (ROCK), participate in a variety of actin-based cellular processes including smooth muscle contraction, cell migration, and stress fiber formation. The ROCK protein consists of an N-terminal kinase domain, a central coiled-coil domain containing a Rho binding site, and a C-terminal pleckstrin homology domain. Here we present the crystal structure of a large section of the central coiled-coil domain of human ROCK I (amino acids 535-700). The structure forms a parallel α-helical coiled-coil dimer that is structurally similar to tropomyosin, an actin filament binding protein. There is an unusual discontinuity in the coiled-coil; three charged residues (E613, R617 and D620) are positioned at what is normally the hydrophobic core of coiled-coil packing. We speculate that this conserved irregularity could function as a hinge that allows ROCK to adopt its autoinhibited conformation.
Organizational Affiliation:
Department of Biological Chemistry and Molecular Pharmacology, Harvard Medical School, Boston, Massachusetts, United States of America.