Structural basis for the substrate specificity of bone morphogenetic protein 1/tolloid-like metalloproteases
Mac Sweeney, A., Parrado, S.G., Vinzenz, D., Bernardi, A., Hein, A., Bodendorf, U., Erbel, P., Logel, C., Gerhartz, B.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Bone morphogenetic protein 1 | 201 | Homo sapiens | Mutation(s): 0 Gene Names: BMP1, PCOLC EC: 3.4.24.19 | 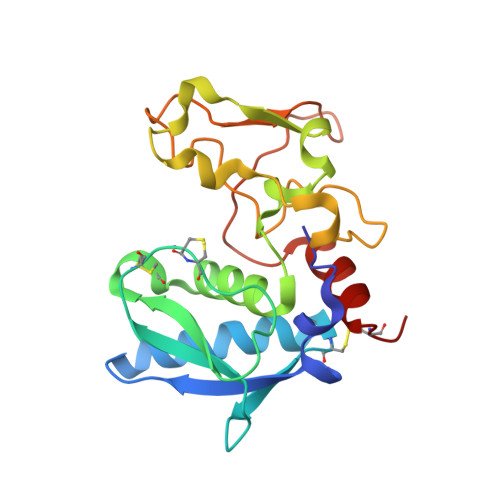 | |
UniProt & NIH Common Fund Data Resources | |||||
Find proteins for P13497 (Homo sapiens) Explore P13497 Go to UniProtKB: P13497 | |||||
PHAROS: P13497 GTEx: ENSG00000168487 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P13497 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| DMS Query on DMS | E [auth A], F [auth A], G [auth A], H [auth A], I [auth A] | DIMETHYL SULFOXIDE C2 H6 O S IAZDPXIOMUYVGZ-UHFFFAOYSA-N |  | ||
| ZN Query on ZN | C [auth A], D [auth A] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| ACE Query on ACE | B [auth A] | ACETYL GROUP C2 H4 O IKHGUXGNUITLKF-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 53.499 | α = 90 |
| b = 59.093 | β = 90 |
| c = 69.451 | γ = 90 |
| Software Name | Purpose |
|---|---|
| XSCALE | data scaling |
| PHASER | phasing |
| REFMAC | refinement |
| PDB_EXTRACT | data extraction |