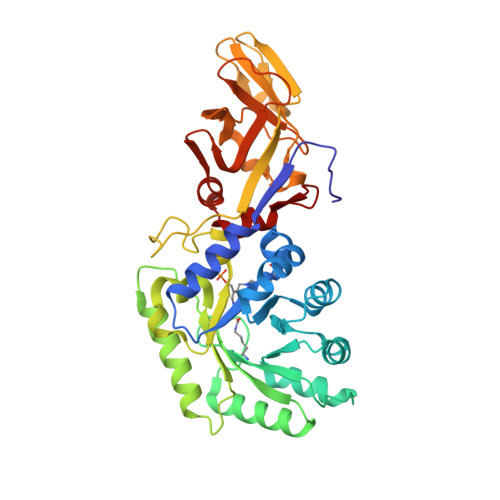
The crystal structure of alanine racemase from Streptococcus pneumoniae, a target for structure-based drug design.
Im, H., Sharpe, M.L., Strych, U., Davlieva, M., Krause, K.L.(2011) BMC Microbiol 11: 116-116
- PubMed: 21612658
- DOI: https://doi.org/10.1186/1471-2180-11-116
- Primary Citation of Related Structures:
3S46 - PubMed Abstract:
Streptococcus pneumoniae is a globally important pathogen. The Gram-positive diplococcus is a leading cause of pneumonia, otitis media, bacteremia, and meningitis, and antibiotic resistant strains have become increasingly common over recent years. Alanine racemase is a ubiquitous enzyme among bacteria and provides the essential cell wall precursor, D-alanine. Since it is absent in humans, this enzyme is an attractive target for the development of drugs against S. pneumoniae and other bacterial pathogens.
Organizational Affiliation:
Research Institute of Pharmaceutical Sciences, College of Pharmacy, Seoul National University, Seoul, Korea.