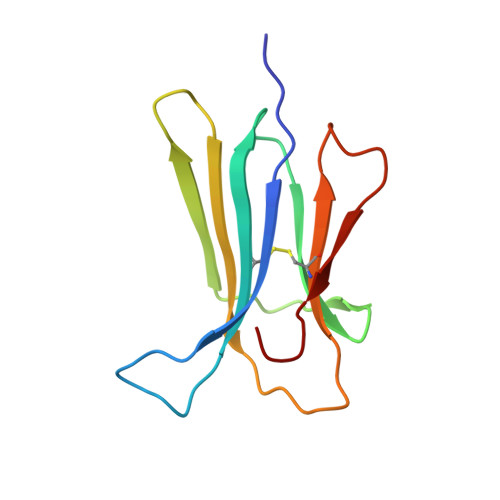
The two tryptophans of beta2-microglobulin have distinct roles in function and folding and might represent two independent responses to evolutionary pressure.
Raimondi, S., Barbarini, N., Mangione, P., Esposito, G., Ricagno, S., Bolognesi, M., Zorzoli, I., Marchese, L., Soria, C., Bellazzi, R., Monti, M., Stoppini, M., Stefanelli, M., Magni, P., Bellotti, V.(2011) BMC Evol Biol 11: 159-159
- PubMed: 21663612
- DOI: https://doi.org/10.1186/1471-2148-11-159
- Primary Citation of Related Structures:
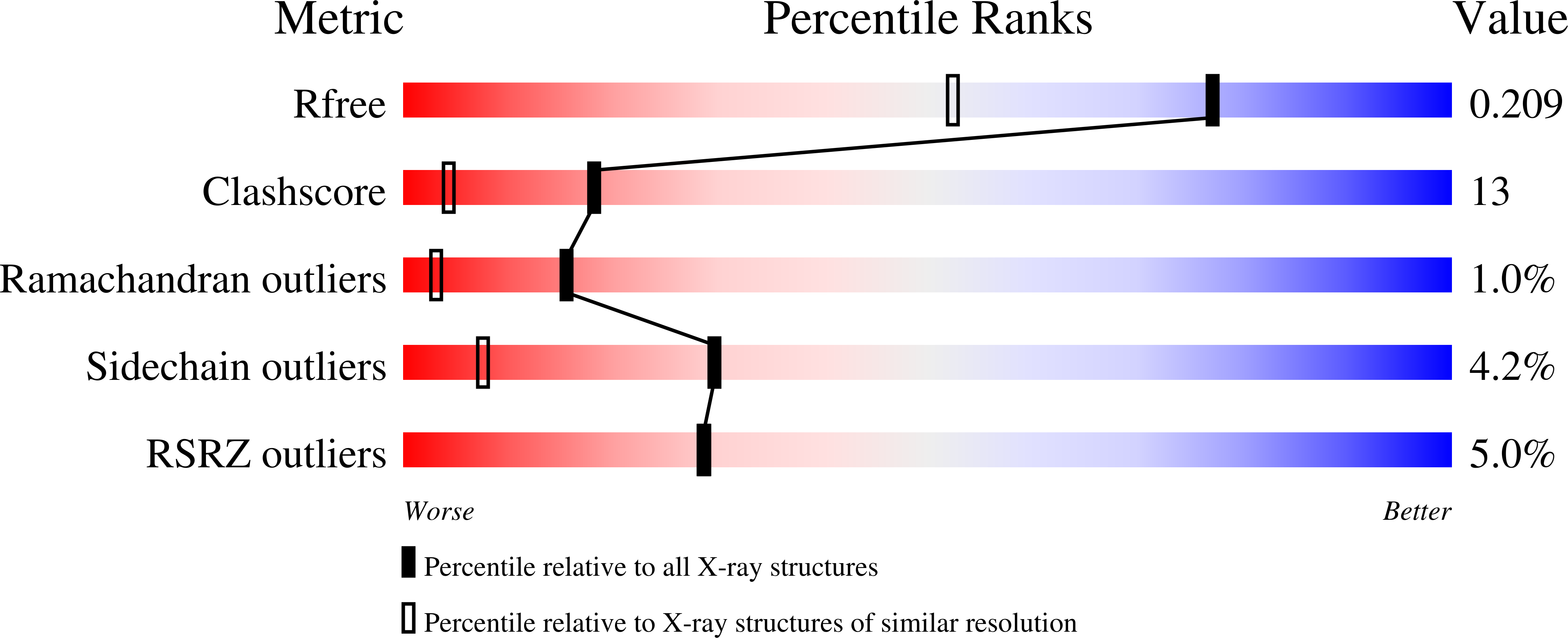
3QDA - PubMed Abstract:
We have recently discovered that the two tryptophans of human β2-microglobulin have distinctive roles within the structure and function of the protein. Deeply buried in the core, Trp95 is essential for folding stability, whereas Trp60, which is solvent-exposed, plays a crucial role in promoting the binding of β2-microglobulin to the heavy chain of the class I major histocompatibility complex (MHCI). We have previously shown that the thermodynamic disadvantage of having Trp60 exposed on the surface is counter-balanced by the perfect fit between it and a cavity within the MHCI heavy chain that contributes significantly to the functional stabilization of the MHCI. Therefore, based on the peculiar differences of the two tryptophans, we have analysed the evolution of β2-microglobulin with respect to these residues.
Organizational Affiliation:
Department of Biochemistry, University of Pavia, via Taramelli 3b, 27100 Pavia, Italy.