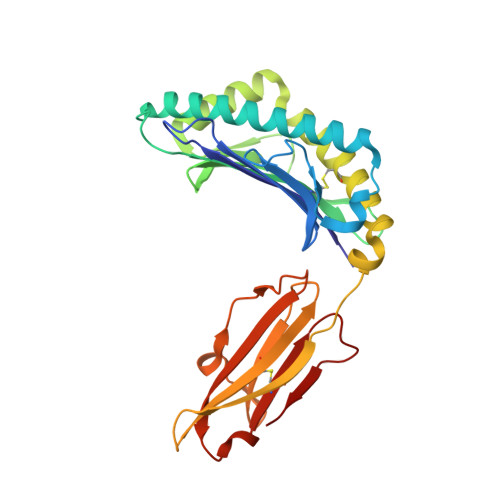
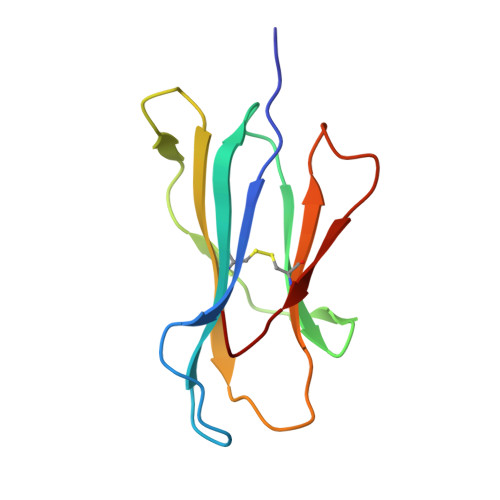
Conformational changes within the HLA-A1:MAGE-A1 complex induced by binding of a recombinant antibody fragment with TCR-like specificity
Kumar, P., Vahedi-Faridi, A., Saenger, W., Ziegler, A., Uchanska-Ziegler, B.(2009) Protein Sci 18: 37-49
- PubMed: 19177349
- DOI: https://doi.org/10.1002/pro.4
- Primary Citation of Related Structures:
3BO8 - PubMed Abstract:
Although there is X-ray crystallographic evidence that the interaction between major histocompatibility complex (MHC, in humans HLA) class I molecules and T cell receptors (TCR) or killer cell Ig-like receptors (KIR) may be accompanied by considerable changes in the conformation of selected residues or even entire loops within TCR or KIR, conformational changes between receptor-bound and -unbound MHC class I molecules of comparable magnitude have not been observed so far. We have previously determined the structure of the MHC class I molecule HLA-A1 bound to a melanoma antigen-encoding gene (MAGE)-A1-derived peptide in complex with a recombinant antibody fragment with TCR-like specificity, Fab-Hyb3. Here, we compare the X-ray structure of HLA-A1:MAGE-A1 with that complexed with Fab-Hyb3 to gain insight into structural changes of the MHC molecule that might be induced by the interaction with the antibody fragment. Apart from the expulsion of several water molecules from the interface, Fab-Hyb3 binding results in major rearrangements (up to 5.5 A) of heavy chain residues Arg65, Gln72, Arg145, and Lys146. Residue 65 is frequently and residues 72 and 146 are occasionally involved in TCR binding-induced conformational changes, as revealed by a comparison with MHC class I structures in TCR-liganded and -unliganded forms. On the other hand, residue 145 is subject to a reorientation following engagement of HLA-Cw4 and KIR2DL1. Therefore, conformational changes within the HLA-A1:MAGE-A1:Fab-Hyb3 complex include MHC residues that are also involved in reorientations in complexes with natural ligands, pointing to their central importance for the peptide-dependent recognition of MHC molecules.
Organizational Affiliation:
Institut für Immungenetik, Charité-Universitätsmedizin Berlin, Campus Benjamin Franklin, Freie Universität Berlin, Thielallee 73, Berlin 14195, Germany.