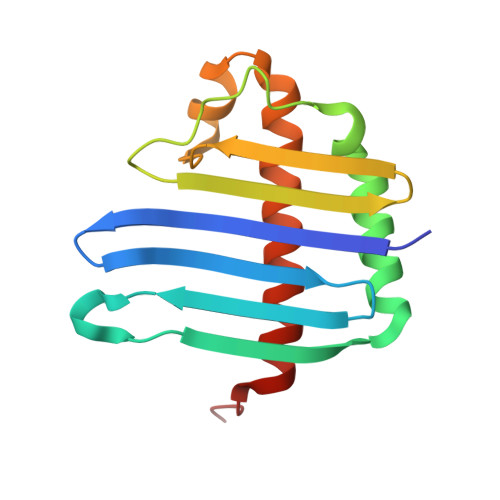
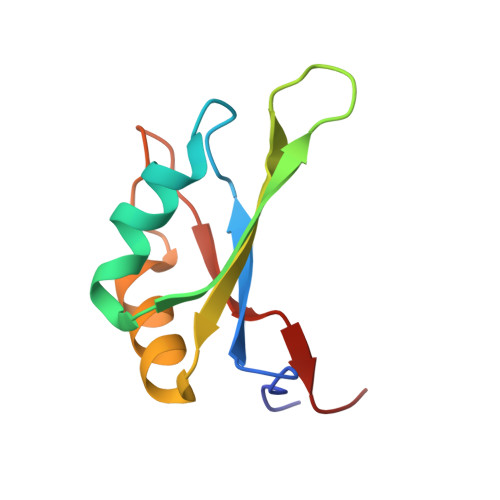
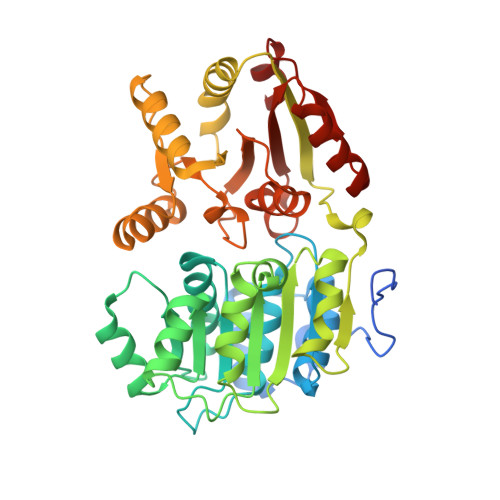
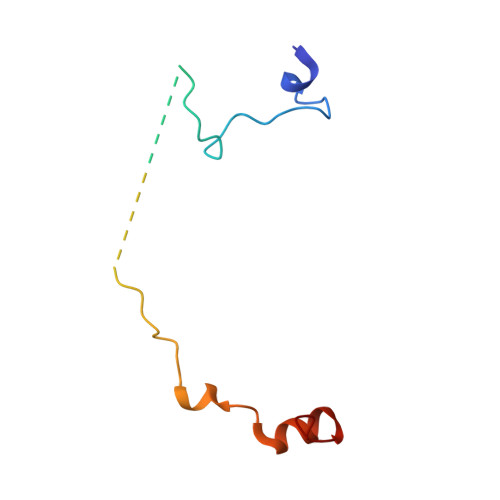
Structure of the exon junction core complex with a trapped DEAD-box ATPase bound to RNA.
Andersen, C.B.F., Ballut, L., Johansen, J.S., Chamieh, H., Nielsen, K.H., Oliveira, C.L., Pedersen, J.S., Seraphin, B., Le Hir, H., Andersen, G.R.(2006) Science 313: 1968-1972
- PubMed: 16931718
- DOI: https://doi.org/10.1126/science.1131981
- Primary Citation of Related Structures:
2HXY, 2HYI - PubMed Abstract:
In higher eukaryotes, a multiprotein exon junction complex is deposited on spliced messenger RNAs. The complex is organized around a stable core, which serves as a binding platform for numerous factors that influence messenger RNA function. Here, we present the crystal structure of a tetrameric exon junction core complex containing the DEAD-box adenosine triphosphatase (ATPase) eukaryotic initiation factor 4AIII (eIF4AIII) bound to an ATP analog, MAGOH, Y14, a fragment of MLN51, and a polyuracil mRNA mimic. eIF4AIII interacts with the phosphate-ribose backbone of six consecutive nucleotides and prevents part of the bound RNA from being double stranded. The MAGOH and Y14 subunits lock eIF4AIII in a prehydrolysis state, and activation of the ATPase probably requires only modest conformational changes in eIF4AIII motif I.
Organizational Affiliation:
Department of Molecular Biology, University of Aarhus, DK-8000 Aarhus, Denmark.