Structure and Inhibition of Orotidine 5'-Monophosphate Decarboxylase from Plasmodium falciparum
Langley, D.B., Shojaei, M., Chan, C., Lok, H.C., Mackay, J.P., Traut, T.W., Guss, J.M., Christopherson, R.I.(2008) Biochemistry 47: 3842-3854
- PubMed: 18303855
- DOI: https://doi.org/10.1021/bi702390k
- Primary Citation of Related Structures:
2ZCG - PubMed Abstract:
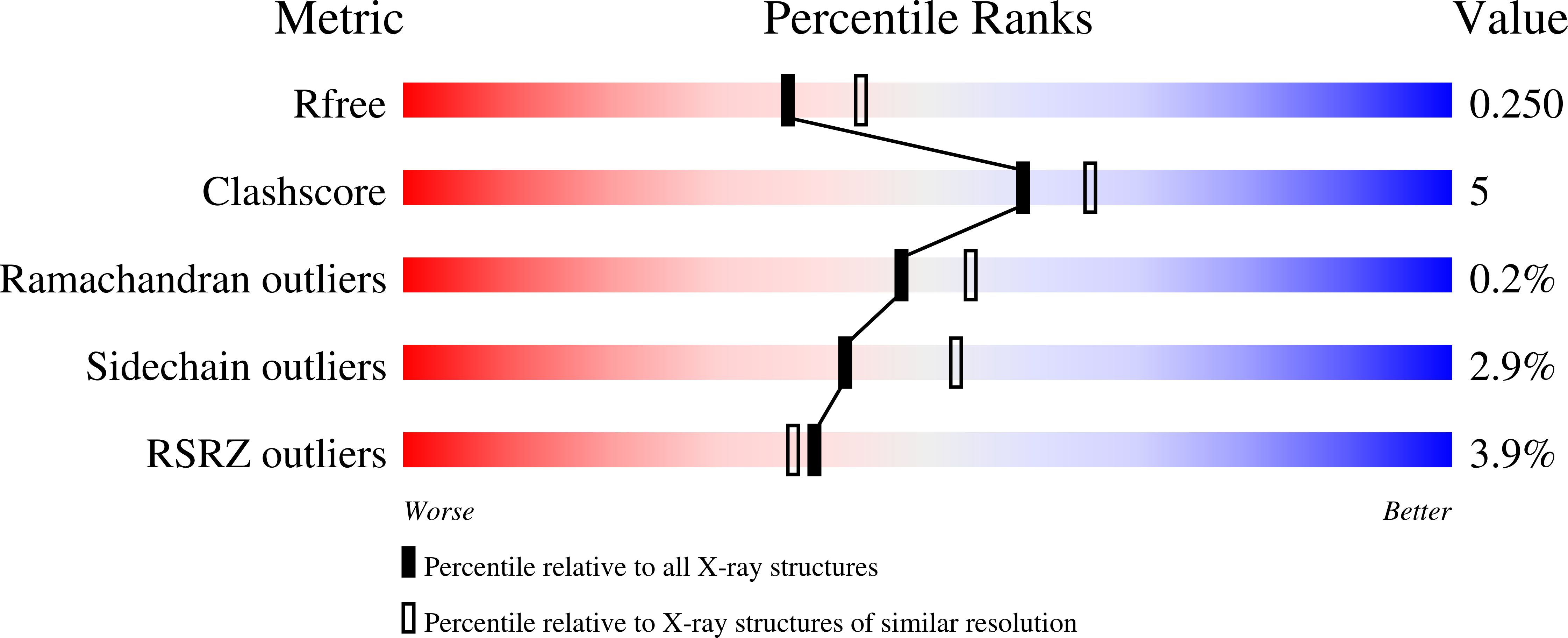
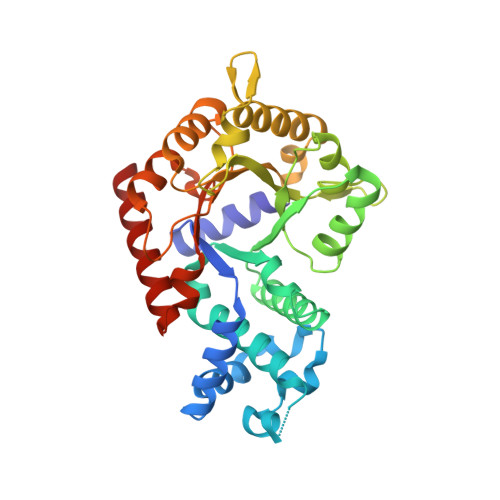
Orotidine 5'-monophosphate (OMP) decarboxylase from Plasmodium falciparum (PfODCase, EC 4.1.1.23) has been overexpressed, purified, subjected to kinetic and biochemical analysis, and crystallized. The native enzyme is a homodimer with a subunit molecular mass of 38 kDa. The saturation curve for OMP as a substrate conformed to Michaelis-Menten kinetics with K m = 350 +/- 60 nM and V max = 2.70 +/- 0.10 micromol/min/mg protein. Inhibition patterns for nucleoside 5'-monophosphate analogues were linear competitive with respect to OMP with a decreasing potency of inhibition of PfODCase in the order: pyrazofurin 5'-monophosphate ( K i = 3.6 +/- 0.7 nM) > xanthosine 5'-monophosphate (XMP, K i = 4.4 +/- 0.7 nM) > 6-azauridine 5'-monophosphate (AzaUMP, K i = 12 +/- 3 nM) > allopurinol-3-riboside 5'-monophosphate ( K i = 240 +/- 20 nM). XMP is an approximately 150-fold more potent inhibitor of PfODCase compared with the human enzyme. The structure of PfODCase was solved in the absence of ligand and displays a classic TIM-barrel fold characteristic of the enzyme. Both the phosphate-binding loop and the betaalpha5-loop have conformational flexibility, which may be associated with substrate capture and product release along the reaction pathway.
Organizational Affiliation:
School of Molecular and Microbial Biosciences, University of Sydney, Sydney, New South Wales 2006, Australia.