High Resolution Structure of Domain III from Class II Fusion Protein of Semliki Forest Virus
Vaney, M.C., Vigouroux, A., Rey, F.A.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| E1 ENVELOPE GLYCOPROTEIN | 91 | Semliki Forest virus | Mutation(s): 0 | 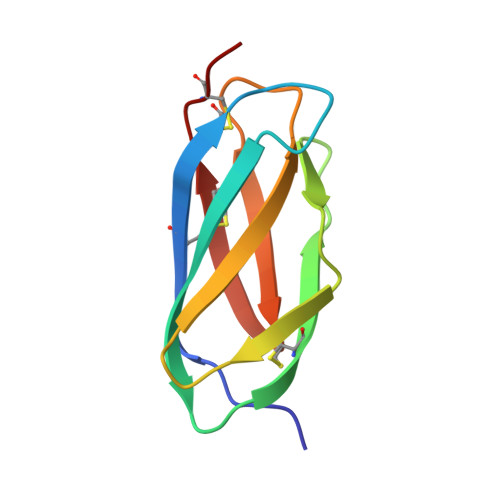 | |
UniProt | |||||
Find proteins for P03315 (Semliki forest virus) Explore P03315 Go to UniProtKB: P03315 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P03315 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| NO3 Query on NO3 | C [auth A], D [auth A] | NITRATE ION N O3 NHNBFGGVMKEFGY-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 60.343 | α = 90 |
| b = 62.659 | β = 90 |
| c = 113.735 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| DENZO | data reduction |
| SCALEPACK | data scaling |
| AMoRE | phasing |