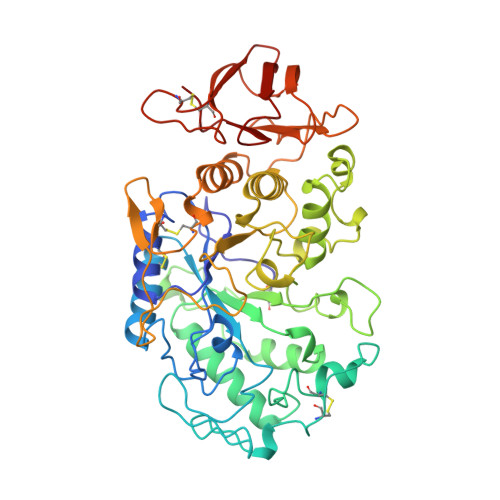
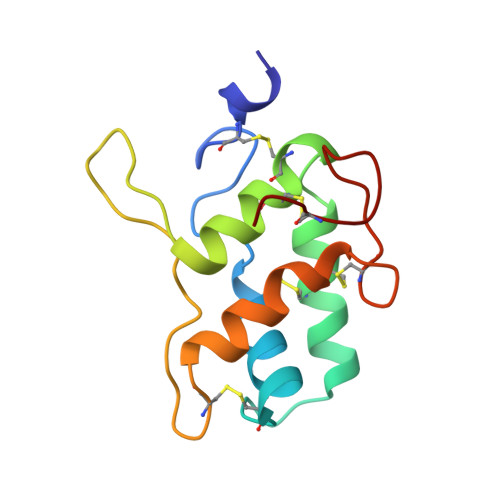
A novel strategy for inhibition of alpha-amylases: yellow meal worm alpha-amylase in complex with the Ragi bifunctional inhibitor at 2.5 A resolution.
Strobl, S., Maskos, K., Wiegand, G., Huber, R., Gomis-Ruth, F.X., Glockshuber, R.(1998) Structure 6: 911-921
- PubMed: 9687373
- DOI: https://doi.org/10.1016/s0969-2126(98)00092-6
- Primary Citation of Related Structures:
1TMQ - PubMed Abstract:
alpha-Amylases catalyze the hydrolysis of alpha-D-(1,4)-glucan linkages in starch and related compounds. There is a wide range of industrial and medical applications for these enzymes and their inhibitors. The Ragi bifunctional alpha-amylase/trypsin inhibitor (RBI) is the prototype of the cereal inhibitor superfamily and is the only member of this family that inhibits both trypsin and alpha-amylases. The mode of inhibition of alpha-amylases by these cereal inhibitors has so far been unknown.
Organizational Affiliation:
Institut für Molekularbiologie und Biophysik, Eidgenössische Technische Hochschule Hönggerberg, Zürich, Switzerland.