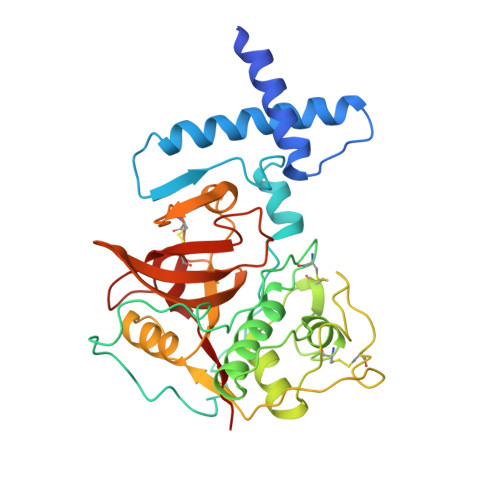
The prosequence of procaricain forms an alpha-helical domain that prevents access to the substrate-binding cleft.
Groves, M.R., Taylor, M.A., Scott, M., Cummings, N.J., Pickersgill, R.W., Jenkins, J.A.(1996) Structure 4: 1193-1203
- PubMed: 8939744
- DOI: https://doi.org/10.1016/s0969-2126(96)00127-x
- Primary Citation of Related Structures:
1PCI - PubMed Abstract:
Cysteine proteases are involved in a variety of cellular processes including cartilage degradation in arthritis, the progression of Alzheimer's disease and cancer invasion: these enzymes are therefore of immense biological importance. Caricain is the most basic of the cysteine proteases found in the latex of Carica papaya. It is a member of the papain superfamily and is homologous to other plant and animal cysteine proteases. Caricain is naturally expressed as an inactive zymogen called procaricain. The inactive form of the protease contains an inhibitory proregion which consists of an additional 106 N-terminal amino acids; the proregion is removed upon activation.
Organizational Affiliation:
Department of Food Macromolecular Science, Institute of Food Research, Reading, UK.