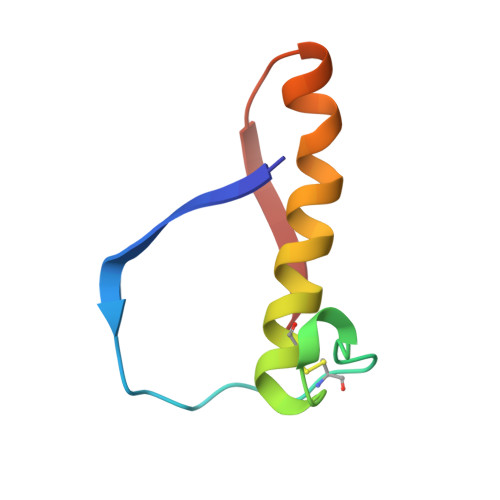
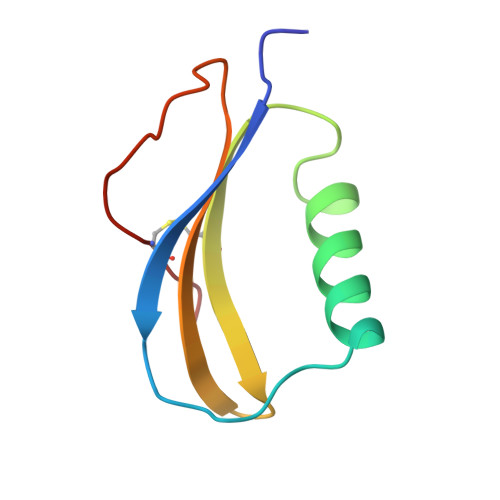
The novel acidophilic structure of the killer toxin from halotolerant yeast demonstrates remarkable folding similarity with a fungal killer toxin.
Kashiwagi, T., Kunishima, N., Suzuki, C., Tsuchiya, F., Nikkuni, S., Arata, Y., Morikawa, K.(1997) Structure 5: 81-94
- PubMed: 9016714
- DOI: https://doi.org/10.1016/s0969-2126(97)00168-8
- Primary Citation of Related Structures:
1KVD, 1KVE - PubMed Abstract:
Several strains of yeasts and fungi produce proteinous substances, termed killer toxins, which kill sensitive strains. The SMK toxin, secreted by the halotolerant yeast Pichia farinosa KK1 strain, uniquely exhibits its maximum killer activity under conditions of acidic pH and high salt concentration. The toxin is composed of two distinct subunits, alpha and beta, which tightly interact with each other under acidic conditions. However, they are easily dissociated under neutral conditions and lose the killer activity. The three-dimensional structure of the SMK toxin will provide a better understanding of the mechanism of toxicity of this protein and the cause of its unique pH-dependent stability.
Organizational Affiliation:
Protein Engineering Research Institute [Biomolecular Engineering Research Institute (BERI) as of the 1st of April 1996], 6-2-3, Furuedai, Suita, Osaka 565, Japan.