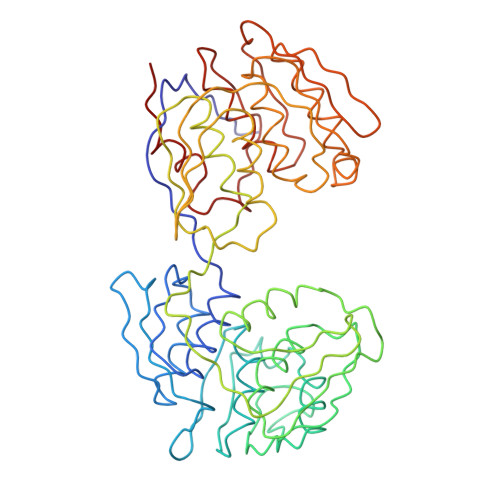
Structure and topological symmetry of the glyphosate target 5-enolpyruvylshikimate-3-phosphate synthase: a distinctive protein fold.
Stallings, W.C., Abdel-Meguid, S.S., Lim, L.W., Shieh, H.S., Dayringer, H.E., Leimgruber, N.K., Stegeman, R.A., Anderson, K.S., Sikorski, J.A., Padgette, S.R., Kishore, G.M.(1991) Proc Natl Acad Sci U S A 88: 5046-5050
- PubMed: 11607190
- DOI: https://doi.org/10.1073/pnas.88.11.5046
- Primary Citation of Related Structures:
1EPS - PubMed Abstract:
5-enol-Pyruvylshikimate-3-phosphate synthase (EPSP synthase; phosphoenolpyruvate:3-phosphoshikimate 1-carboxyvinyltransferase, EC 2.5.1.19) is an enzyme on the pathway toward the synthesis of aromatic amino acids in plants, fungi, and bacteria and is the target of the broad-spectrum herbicide glyphosate. The three-dimensional structure of the enzyme from Escherichia coli has been determined by crystallographic techniques. The polypeptide backbone chain was traced by examination of an electron density map calculated at 3-A resolution. The two-domain structure has a distinctive fold and appears to be formed by 6-fold replication of a protein folding unit comprising two parallel helices and a four-stranded sheet. Each domain is formed from three of these units, which are related by an approximate threefold symmetry axis; in each domain three of the helices are completely buried by a surface formed from the three beta-sheets and solvent-accessible faces of the other three helices. The domains are related by an approximate dyad, but in the present crystals the molecule does not display pseudo-symmetry related to the symmetry of point group 32 because its approximate threefold axes are almost normal. A possible relation between the three-dimensional structure of the protein and the linear sequence of its gene will be described. The topological threefold symmetry and orientation of each of the two observed globular domains may direct the binding of substrates and inhibitors by a helix macrodipole effect and implies that the active site is located near the interdomain crossover segments. The structure also suggests a rationale for the glyphosate tolerance conferred by sequence alterations.
Organizational Affiliation:
Monsanto Corporate Research and Technology Division of Monsanto Agricultural Company, Monsanto Company, St. Louis, MO 63198, USA.