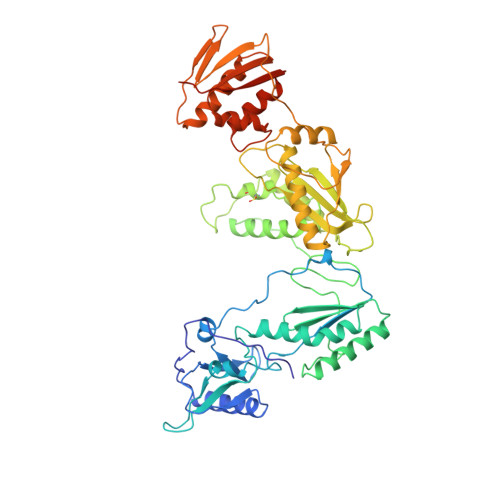
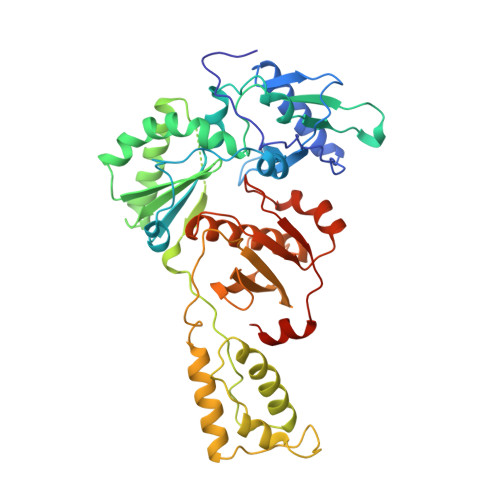
The structure of HIV-1 reverse transcriptase complexed with 9-chloro-TIBO: lessons for inhibitor design.
Ren, J., Esnouf, R., Hopkins, A., Ross, C., Jones, Y., Stammers, D., Stuart, D.(1995) Structure 3: 915-926
- PubMed: 8535785
- DOI: https://doi.org/10.1016/S0969-2126(01)00226-X
- Primary Citation of Related Structures:
1REV - PubMed Abstract:
HIV reverse transcriptase (RT) is a key target of anti-AIDS therapies. Structural studies of HIV-1 RT, unliganded and complexed with different non-nucleoside inhibitors (NNIs), have pointed to a common mode of binding and inactivation through distortion of the polymerase catalytic site by NNIs containing two hinged rings. The mode of binding of the TIBO family of inhibitors is of interest because these compounds do not fit the two-hinged-ring model.
Organizational Affiliation:
Laboratory of Molecular Biophysics, Oxford, UK.