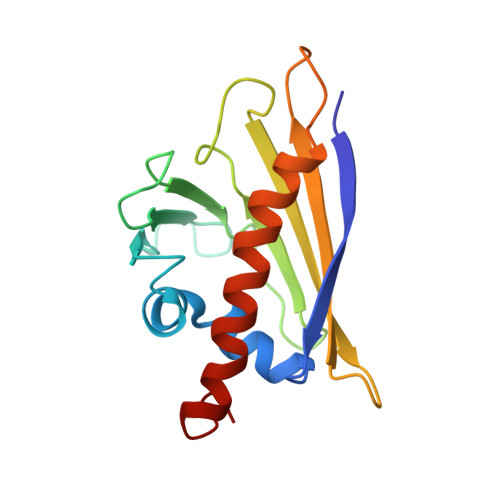
X-ray and NMR structure of Bet v 1, the origin of birch pollen allergy.
Gajhede, M., Osmark, P., Poulsen, F.M., Ipsen, H., Larsen, J.N., Joost van Neerven, R.J., Schou, C., Lowenstein, H., Spangfort, M.D.(1996) Nat Struct Biol 3: 1040-1045
- PubMed: 8946858
- DOI: https://doi.org/10.1038/nsb1296-1040
- Primary Citation of Related Structures:
1BTV, 1BV1 - PubMed Abstract:
The three-dimensional structure of the major birch pollen allergen, the 17,500 M(r) acidic protein Bet v 1 (from the birch, Betula verrucosa), is presented as determined both in the crystalline state by X-ray diffraction and in solution by nuclear magnetic resonance (NMR) spectroscopy. This is the first experimentally determined structure of a clinically important inhalant major allergen, estimated to cause allergy in 5-10 million individuals worldwide. The structure shows three regions on the molecular surface predicted to harbour cross-reactive B-cell epitopes which provide a structural basis for the allergic symptoms that birch pollen allergic patients show when they encounter pollens from related trees such as hazel, alder and hornbeam. The structure also shows an unusual feature, a 30 A-long forked cavity that penetrates the entire protein.
Organizational Affiliation:
Department of Chemistry, University of Copenhagen, Denmark.