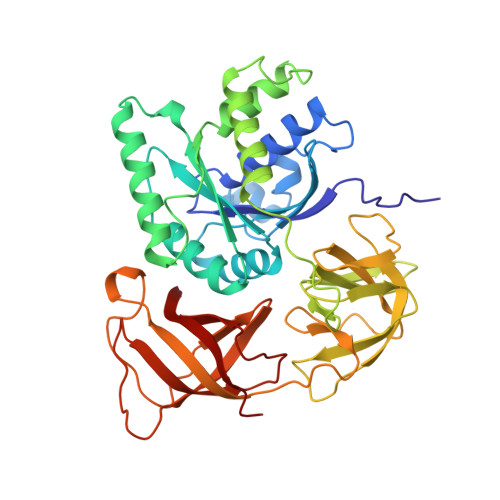
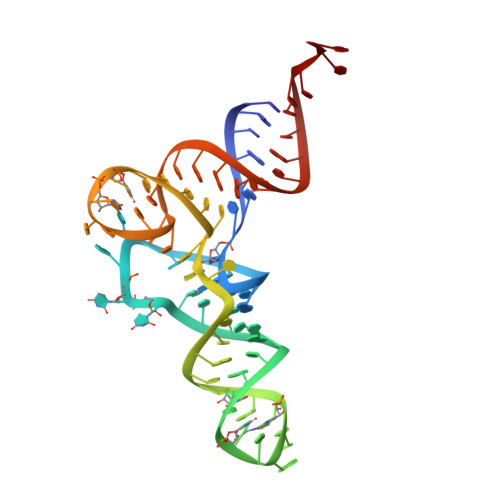
The crystal structure of Cys-tRNACys-EF-Tu-GDPNP reveals general and specific features in the ternary complex and in tRNA.
Nissen, P., Thirup, S., Kjeldgaard, M., Nyborg, J.(1999) Structure 7: 143-156
- PubMed: 10368282
- DOI: https://doi.org/10.1016/s0969-2126(99)80021-5
- Primary Citation of Related Structures:
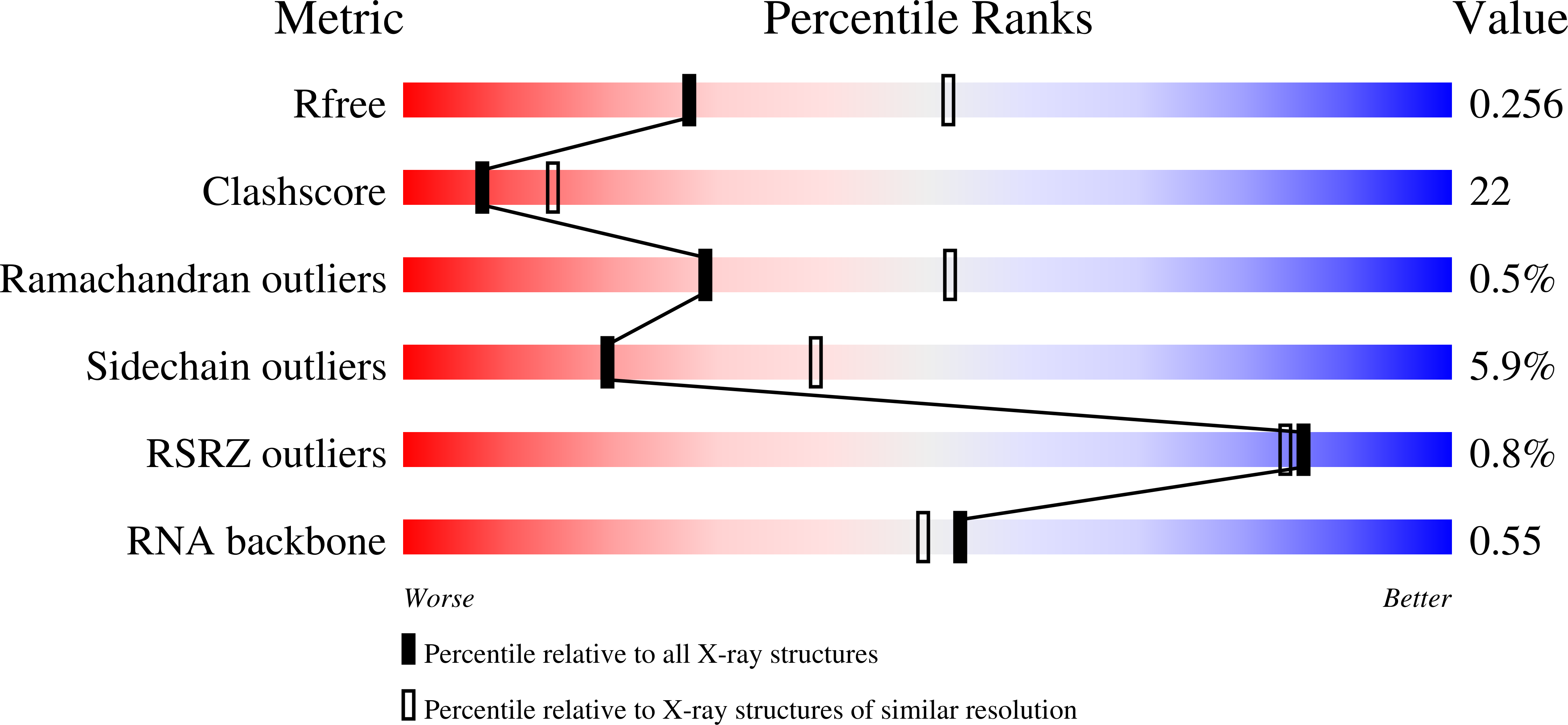
1B23 - PubMed Abstract:
. The translation elongation factor EF-Tu in its GTP-bound state forms a ternary complex with any aminoacylated tRNA (aa-tRNA), except initiator tRNA and selenocysteinyl-tRNA. This complex delivers aa-tRNA to the ribosomal A site during the elongation cycle of translation. The crystal structure of the yeast Phe-tRNAPhe ternary complex with Thermus aquaticus EF-Tu-GDPNP (Phe-TC) has previously been determined as one representative of this general yet highly discriminating complex formation.
Organizational Affiliation:
Institute of Molecular and Structural Biology, Aarhus University, Gustav Wieds Vej 10, C DK 8000 Aarhus C, Denmark.